EMSA vs Gel Retardation Assay: A Complete Guide to Electrophoretic Mobility Shift Assays for Researchers
This article provides a comprehensive guide for researchers and drug development professionals on Electrophoretic Mobility Shift Assays (EMSA), clarifying its relationship to the broader term 'gel retardation assay.' We cover...
EMSA vs Gel Retardation Assay: A Complete Guide to Electrophoretic Mobility Shift Assays for Researchers
Abstract
This article provides a comprehensive guide for researchers and drug development professionals on Electrophoretic Mobility Shift Assays (EMSA), clarifying its relationship to the broader term 'gel retardation assay.' We cover the foundational principles and historical context, detail modern methodological protocols and applications in gene regulation and drug discovery, offer advanced troubleshooting and optimization strategies, and present a critical comparative analysis with alternative validation techniques. Our goal is to equip scientists with the knowledge to implement robust, publication-quality nucleic acid-protein interaction studies.
Understanding EMSA: The Core Principles and History Behind the Gel Retardation Assay
This whitepaper, framed within a thesis on differentiating EMSA and gel retardation assay research, provides a technical dissection of these synonymous methodologies. The Electrophoretic Mobility Shift Assay (EMSA), interchangeably termed gel shift or gel retardation assay, is a cornerstone technique in molecular biology for detecting protein-nucleic acid interactions. It is pivotal for research into gene regulation, drug discovery targeting transcription factors, and understanding pathological mechanisms.
Core Principle and Definitions
The fundamental principle relies on the reduction in electrophoretic mobility of a nucleic acid probe when bound by a protein, causing a "shift" or "retardation" in its migration through a native polyacrylamide or agarose gel. Despite the synonymous use of terms, some research contexts make subtle distinctions, as explored in this thesis.
- EMSA (Electrophoretic Mobility Shift Assay): The most precise and widely adopted term, emphasizing the measurement of mobility change.
- Gel Shift Assay: A common shorthand, highlighting the visual shift of the probe band.
- Gel Retardation Assay: An older, descriptive term focusing on the retarded migration of the complex.
Experimental Protocol: A Standard EMSA Workflow
1. Probe Preparation:
- Design: Chemically synthesize or PCR-amplify a 20-40 bp DNA/RNA sequence containing the suspected protein-binding site.
- Labeling: End-label the probe with gamma-³²P-ATP using T4 Polynucleotide Kinase or use a fluorescent/chemiluminescent tag for non-radioactive detection. Purify using a spin column.
2. Protein Sample Preparation:
- Source nuclear extracts, purified recombinant proteins, or whole-cell lysates.
- Pre-clear extracts to remove non-specific binding components.
3. Binding Reaction:
- Assemble in a 10-20 µL volume:
- Labeled probe: 10,000-20,000 cpm (or ~1-10 fmol).
- Protein extract: 2-20 µg of nuclear extract or 10-100 ng of purified protein.
- Poly(dI-dC): 1-2 µg, as a non-specific competitor.
- Buffer: 10 mM HEPES, 50 mM KCl, 1 mM DTT, 2.5% glycerol, 0.05% NP-40, pH 7.9.
- Incubate at 20-25°C for 20-30 minutes.
4. Electrophoresis:
- Load samples onto a pre-run, native 4-10% polyacrylamide gel (29:1 acrylamide:bis) in 0.5X TBE buffer at 4-10°C.
- Run at constant voltage (100-150 V) until the free probe nears the bottom.
5. Detection:
- For radioactive probes: Dry gel and expose to a Phosphorimager screen or X-ray film.
- For fluorescent probes: Image directly using a suitable scanner.
Data Presentation and Validation
Table 1: Key Controls for EMSA Experiment Validation
| Control Type | Purpose | Expected Result |
|---|---|---|
| Free Probe | Baseline mobility of unbound nucleic acid. | Single, fast-migrating band. |
| Protein + Probe | Detection of specific complex formation. | Additional, slower-migrating band(s). |
| Specific Competitor (unlabeled wild-type probe) | Confirms binding specificity via competition. | Disappearance or reduction of shifted band. |
| Non-specific Competitor (unlabeled mutant probe) | Confirms sequence specificity of interaction. | No reduction in shifted band intensity. |
| Antibody Supershift | Identifies specific protein in complex. | Further retardation or loss of shifted band. |
| Mutation Probe | Maps critical binding sequence. | Absence of shifted band with mutant probe. |
Table 2: Quantitative Data from a Representative EMSA Experiment
| Condition | % Free Probe | % Shifted Complex | Relative Binding Affinity (Arbitrary Units) |
|---|---|---|---|
| Free Probe Only | 99.8 | 0.2 | - |
| Protein + Probe | 45.3 | 54.7 | 1.00 |
| + 50x Cold Competitor | 92.1 | 7.9 | 0.14 |
| + 50x Mutant Competitor | 48.5 | 51.5 | 0.94 |
| + Specific Antibody | 46.2 | 38.1 (Supershift: 15.7) | N/A |
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Function in EMSA |
|---|---|
| T4 Polynucleotide Kinase | Catalyzes the transfer of a radioactive phosphate group to the 5' end of the nucleic acid probe. |
| Poly(dI-dC) | A synthetic, non-specific nucleic acid polymer used as a competitor to absorb non-specific DNA/RNA-binding proteins. |
| HEPES/KCl Binding Buffer | Maintains optimal ionic strength and pH to facilitate specific protein-nucleic acid interactions during incubation. |
| Native Polyacrylamide Gel | A non-denaturing gel matrix that separates protein-nucleic acid complexes based on size, charge, and shape. |
| Non-radioactive Labeling Kits (Biotin/DIG) | Provide safer alternatives to radioisotopes for probe labeling, utilizing chemiluminescence or fluorescence for detection. |
| Phosphorimager Screen & Scanner | Essential for high-sensitivity, quantitative digital imaging of radioactive or fluorescent gels. |
Methodological Nuances and Thesis Context
Within the broader thesis, a critical distinction is drawn between classic EMSA and specific implementations of gel retardation assays. EMSA typically implies a focus on specific, sequence-defined binding events, confirmed through rigorous competition and supershift controls. In contrast, some "gel retardation" literature may describe assays measuring non-specific or aggregate nucleic acid binding, often using different gel matrices (e.g., agarose for larger complexes) and fewer specificity controls. This distinction is crucial for accurate data interpretation in drug development, where targeting specific transcription factor interactions is the goal.
Title: EMSA Standard Experimental Workflow
Title: EMSA Result Interpretation Logic
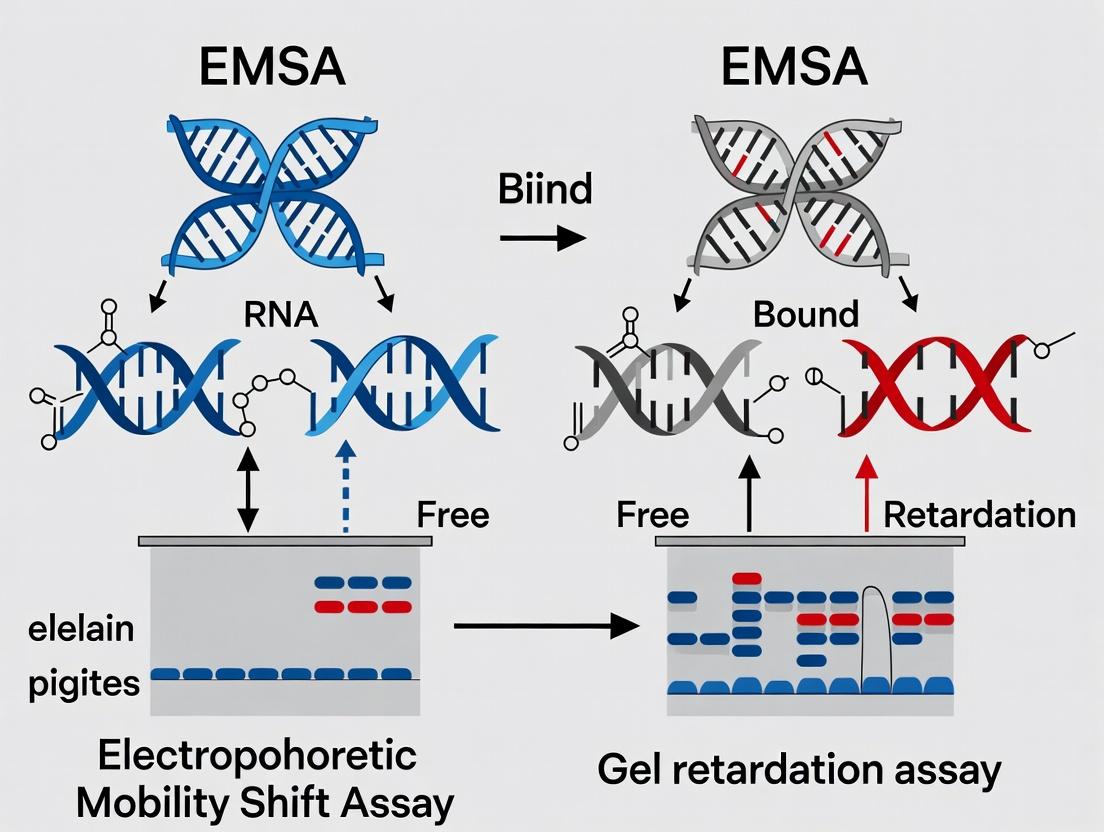
The Electrophoretic Mobility Shift Assay (EMSA), also interchangeably termed the gel retardation or band shift assay, is a cornerstone technique in molecular biology for studying protein-nucleic acid interactions. While the terms are often used synonymously, a nuanced thesis distinguishes EMSA as a specific application of the broader gel retardation principle. The core principle underlying both is that the formation of a complex between a protein and a nucleic acid probe (DNA or RNA) results in a detectable reduction (retardation) of the probe's electrophoretic mobility through a non-denaturing polyacrylamide or agarose gel. This "shift" manifests as a band that migrates more slowly than the free probe. The degree of retardation is influenced by the size, charge, and conformational changes imparted by the protein binding event. This whitepaper delves into the biophysical and technical foundations of this shift, providing a detailed guide for researchers applying this principle in gene regulation studies, drug discovery, and diagnostic development.
Biophysical Basis of the Shift: Charge, Mass, and Conformation
The migration of a nucleic acid through a gel matrix during electrophoresis is governed by its net charge, mass/size, and three-dimensional shape. Protein binding alters all three parameters.
- Charge Masking: Nucleic acids possess a uniform negative charge per phosphate group. DNA-binding proteins often carry positively charged amino acid residues (e.g., lysine, arginine) that interact with the sugar-phosphate backbone. This interaction partially neutralizes the net negative charge of the complex, reducing the electrophoretic force pulling it toward the anode.
- Increased Mass: The addition of protein mass increases the hydrodynamic volume of the complex. Larger entities experience greater frictional resistance from the gel matrix, slowing migration.
- Conformational Change: Protein binding can induce bending, looping, or kinking of the nucleic acid. This altered conformation can increase the effective molecular radius, causing further retardation beyond that predicted by simple mass addition.
The combined effect is a quantifiable upward shift in the band position relative to the free probe. Supershift assays, which incorporate a specific antibody, add a third layer of mass and charge, causing a further retardation and confirming the identity of the bound protein.
Table 1: Impact of Protein Binding Parameters on Electrophoretic Mobility
| Parameter Altered by Protein Binding | Effect on Complex | Result on Gel Migration | Approximate % Retardation Range (Typical) |
|---|---|---|---|
| Net Negative Charge | Decreased | Slower | 5-20% (Primary contributor) |
| Hydrodynamic Radius / Mass | Increased | Slower | 10-30% (Size-dependent) |
| Nucleic Acid Conformation | Bent/Kinked | Significantly Slower | 15-50% (e.g., HMG-box proteins) |
| Multimerization State | Increased (e.g., dimer) | Much Slower | 25-60% |
Table 2: Comparative Analysis: EMSA vs. Broader Gel Retardation Assay
| Feature | Classic EMSA (Thesis Context) | Broader Gel Retardation Principle |
|---|---|---|
| Primary Objective | Detect specific protein-nucleic acid complexes. | Detect any complex that alters mobility. |
| Typical Probe | Short, defined DNA/RNA sequence (20-50 bp). | Can be large plasmids, PCR products, or RNA. |
| Gel Matrix | Non-denaturing polyacrylamide (high resolution). | Non-denaturing agarose or polyacrylamide. |
| Quantification Focus | Binding affinity (Kd), stoichiometry, specificity. | Presence/absence of binding, complex size. |
| Common Applications | Transcription factor studies, miRNA-protein. | Ribonucleoprotein complexes, drug-DNA intercalation. |
Detailed Experimental Protocol: A Standard EMSA
Protocol: Transcription Factor EMSA using a 32P-labeled DNA Probe
Objective: To detect and characterize the binding of a nuclear extract protein to its cognate DNA consensus sequence.
Part A: Probe Preparation and Labeling (End-Labeling)
- Annealing: Mix complementary single-stranded oligonucleotides (containing the protein-binding site) in equimolar ratios in annealing buffer (10 mM Tris, 50 mM NaCl, 1 mM EDTA). Heat to 95°C for 5 min and slowly cool to room temperature.
- Phosphorylation: In a 20 µL reaction, combine:
- 100 ng double-stranded oligonucleotide.
- 2 µL T4 Polynucleotide Kinase 10X Buffer.
- 20 µCi [γ-³²P] ATP.
- 10 U T4 Polynucleotide Kinase.
- Nuclease-free water to volume.
- Incubate at 37°C for 45 min.
- Purification: Remove unincorporated nucleotides using a spin column (e.g., G-25 Sephadex) or ethanol precipitation. Resuspend in TE buffer. Determine specific activity by scintillation counting.
Part B: Binding Reaction
- Set up reactions on ice (20 µL final volume):
- 1 µL 32P-labeled probe (~20,000 cpm).
- 2 µL 10X Binding Buffer (100 mM Tris pH 7.5, 500 mM NaCl, 50% glycerol, 10 mM DTT, 10 mM EDTA).
- 1 µL Poly(dI-dC) (1 µg/µL) as non-specific competitor.
- X µL Nuclear extract or purified protein (typically 2-10 µg).
- Y µL Nuclease-free water.
- For competition: Add 100-fold molar excess of unlabeled specific or mutant cold probe.
- For supershift: Add 1-2 µg of specific antibody.
- Mix gently and incubate at room temperature for 25-30 min.
Part C: Non-Denaturing Gel Electrophoresis
- Prepare a 6% non-denaturing polyacrylamide gel (29:1 acrylamide:bis) in 0.5X TBE buffer. Pre-run the gel at 100 V for 60 min in a cold room (4-10°C).
- Add 5 µL of 5X loading dye (glycerol, bromophenol blue) to each binding reaction. Do not heat.
- Load samples onto the pre-run gel. Run at 100 V in 0.5X TBE at 4°C until the dye front is near the bottom (≈1.5-2 hrs).
- Transfer gel to blotting paper, dry under vacuum, and expose to a phosphorimager screen overnight. Analyze using imaging software.
Visualization: EMSA Workflow and Principle
Title: EMSA Experimental Workflow from Binding to Detection
Title: Core Principle of Electrophoretic Retardation Upon Protein Binding
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents for EMSA/Gel Retardation Assays
| Reagent / Material | Function / Purpose | Critical Notes |
|---|---|---|
| Purified Protein or Nuclear/Cytoplasmic Extract | Source of the nucleic acid-binding protein. | Extract quality is paramount; use fresh protease inhibitors. |
| Labeled Nucleic Acid Probe (³²P, Digoxigenin, Fluorescent, Biotin) | Target for binding; enables detection. | ³²P offers high sensitivity; non-radioactive alternatives are safer. |
| Non-Specific Competitor DNA (Poly(dI-dC), sheared salmon sperm DNA) | Binds to non-specific proteins, reducing background. | Concentration must be optimized for each protein-probe pair. |
| 10X Binding Buffer (with glycerol, salt, DTT, carrier) | Provides optimal ionic strength and reducing environment for binding. | Glycerol (10-20%) aids in loading. DTT maintains protein redux state. |
| Non-Denaturing Polyacrylamide Gel (4-10%) | Sieving matrix that separates based on size, charge, and shape. | Acrylamide concentration determines resolution range. Must be run cold. |
| Specific Unlabeled "Cold" Competitor Probe | Confirms binding specificity via competition. | 50-200x molar excess typical. Mutant probe validates sequence specificity. |
| Specific Antibody (for Supershift) | Binds to protein in complex, causing a further mobility shift. | Confirms protein identity. Control: non-specific IgG. |
| Electrophoresis Buffer (0.5X TBE or TAE) | Conducts current and maintains pH during separation. | Low ionic strength buffers (0.5X) are standard; run at constant voltage. |
| Detection System (Phosphorimager, X-ray film, Chemiluminescence imager) | Visualizes the separated probe and complexes. | Choice depends on probe label. Phosphorimagers offer quantitative analysis. |
The study of DNA-protein interactions is fundamental to understanding gene regulation. This whitepaper traces the historical progression of key methodologies leading to the modern Electrophoretic Mobility Shift Assay (EMSA), contextualized within the broader research thesis distinguishing EMSA from the more generic term "gel retardation assay." While often used interchangeably, "gel retardation assay" describes the core phenomenon, whereas "EMSA" represents a specific, optimized, and widely adopted technical implementation of that principle.
Historical Progression and Key Discoveries
The journey began with the conceptual understanding that proteins bind to specific DNA sequences to control transcription. The development of filter binding assays in the 1970s provided early quantitative data but lacked resolution for complex mixtures. The pivotal shift occurred with the introduction of gel-based separation, which offered superior resolution to detect specific complexes.
Table 1: Historical Milestones in DNA-Protein Interaction Analysis
| Year | Milestone | Key Innovation | Limitation Overcome | |
|---|---|---|---|---|
| 1972 | Filter Binding Assay (Riggs et al.) | Nitrocellulose filter retains protein-bound DNA. | First quantitative measure of binding constants. | Low resolution; cannot distinguish multiple complexes. |
| 1981 | Gel Retardation Assay (Garner & Revzin, Fried & Crothers) | Native polyacrylamide gel electrophoresis separates free and protein-bound DNA. | Visual resolution of specific vs. non-specific complexes. | Low sensitivity; large amounts of protein and DNA required. |
| 1986 | Electrophoretic Mobility Shift Assay (EMSA) term popularized | Optimization of gel conditions (pH, ionic strength, glycerol). | Enhanced complex stability during electrophoresis. | Established standard protocol for reproducibility. |
| 1988-90 | Supershift & Competition EMSA | Use of specific antibodies (supershift) or unlabeled competitor DNA. | Identifies protein component & assesses binding specificity. | Antibody must not disrupt the protein-DNA interface. |
| Modern | Fluorescence-based & Capillary EMSA | Fluorescent DNA labels & capillary electrophoresis detection. | Quantitative, high-throughput, and reduced assay time. | Requires specialized instrumentation. |
Core Methodology: Modern EMSA Protocol
The following is a detailed protocol for a standard, non-radioactive EMSA.
1. Probe Preparation:
- Labeling: A 20-50 bp double-stranded DNA probe containing the suspected protein-binding site is prepared. The probe is typically labeled at the 5' end using T4 polynucleotide kinase and [γ-32P]ATP (radioactive) or a fluorophore-tagged nucleotide (non-radioactive, e.g., Cy5). Purify the labeled probe using a spin column.
- Annealing: If using single-stranded oligonucleotides, combine equimolar amounts of complementary strands in 1X annealing buffer (10 mM Tris, 50 mM NaCl, 1 mM EDTA, pH 7.5-8.0), heat to 95°C for 5 minutes, and cool slowly to room temperature.
2. Binding Reaction:
- Assemble a 20 µL reaction mix on ice:
- 1X Binding Buffer (10 mM HEPES, 50 mM KCl, 1 mM DTT, 2.5 mM MgCl2, 0.05% NP-40, pH 7.9).
- 1 µg Poly(dI·dC) or other non-specific carrier DNA.
- 0.5-2 µg nuclear extract or 1-100 nM purified protein.
- Labeled DNA probe (10-20 fmol).
- Critical Controls: Include reactions with a 100-fold molar excess of unlabeled specific competitor (to demonstrate specificity) and non-specific competitor (e.g., mutated sequence). For supershift, add 1-2 µg of specific antibody after the initial binding and incubate further.
- Incubate at 20-25°C for 20-30 minutes.
3. Electrophoresis:
- Pre-run a 4-6% native polyacrylamide gel (29:1 acrylamide:bis) in 0.5X TBE (or TAE) buffer at 4-10°C for 30-60 min. The low cross-linker ratio and cold temperature help maintain complexes.
- Load samples (without loading dye, which can disrupt complexes) directly. Load a separate lane with dye-only for tracking.
- Run at 80-100 V, constant voltage, for 1.5-2 hours, maintaining cold temperature (4°C).
4. Detection:
- For radioactive probes: Dry gel and expose to a phosphorimager screen.
- For fluorescent probes: Image directly using a fluorescence scanner with appropriate excitation/emission settings.
- For chemiluminescent probes (biotinylated): Transfer to nylon membrane, UV crosslink, and develop with streptavidin-HRP.
Diagram Title: Modern EMSA Experimental Workflow and Output
The Scientist's Toolkit: Key EMSA Reagent Solutions
Table 2: Essential Research Reagents for EMSA
| Reagent/Solution | Function & Rationale |
|---|---|
| Labeled DNA Probe | Contains the specific sequence of interest; the label (radioactive, fluorescent) enables detection. |
| Nuclear Extract | Source of transcription factors and DNA-binding proteins; prepared from cells of interest. |
| Poly(dI·dC) | Non-specific competitor DNA; blocks non-specific protein interactions with the probe or tube. |
| Specific Unlabeled Competitor | Excess identical unlabeled DNA; validates binding specificity by competing away the shift. |
| Binding Buffer | Provides optimal ionic strength, pH, and co-factors (e.g., Mg2+, DTT) for protein-DNA interaction. |
| Native Polyacrylamide Gel | Matrix for separation based on size/charge of complex without denaturing components. |
| Antibody (for Supershift) | Antibody specific to the DNA-binding protein; binds complex causing a further mobility reduction ("supershift"). |
Quantitative Data & Comparison
Modern quantitative EMSA (Q-EMSA) allows determination of binding affinities (Kd) and stoichiometry.
Table 3: Quantitative EMSA Data Analysis Parameters
| Parameter | How it's Derived from EMSA | Typical Range for a Tight Complex | Notes |
|---|---|---|---|
| Apparent Kd | Fraction bound vs. protein concentration curve (via densitometry). | 0.1 - 10 nM | Assumes equilibrium is maintained during gel run. |
| Stoichiometry | Mole ratio of protein to DNA at complete shift; or by molecular weight analysis of complex. | 1:1, 2:1, or higher order. | Supershift can confirm protein identity. |
| Binding Specificity | IC50 from competition curves with labeled vs. unlabeled specific/non-specific DNA. | >100-fold difference in IC50. | Core validation control. |
EMSA vs. Gel Retardation Assay: A Thesis Context
Within the broader research thesis, the distinction is critical. The Gel Retardation Assay is the foundational principle—any assay where a biomolecular interaction causes a decreased electrophoretic mobility. EMSA is a refined subset, characterized by:
- Standardized buffer systems to preserve non-covalent complexes.
- Defined gel compositions (low cross-linker, native conditions).
- Systematic use of critical controls (competition, supershift).
- The capability for quantitative analysis.
Thus, all EMSAs are gel retardation assays, but not all gel retardation assays achieve the specific, controlled, and quantitatively analyzable standard of a modern EMSA. This evolution from a qualitative gel shift to a robust, quantitative EMSA marks the maturation of the technique into a cornerstone of molecular biology and drug discovery, where it is used to screen compounds that modulate transcription factor activity.
The Electrophoretic Mobility Shift Assay (EMSA), often synonymously termed the gel retardation assay, is a cornerstone technique in molecular biology for detecting protein-nucleic acid interactions. Within the broader thesis of differentiating EMSA from the more general concept of a gel retardation assay, it is critical to understand that EMSA specifically refers to the use of a non-denaturing gel to resolve complexes formed between a nucleic acid probe and components within a protein extract. The precise preparation and quality of these three core components directly determine the assay's specificity, sensitivity, and validity. This guide details their optimal preparation and integration.
Core Component 1: The Nucleic Acid Probe
The probe is the labeled nucleic acid sequence containing the putative protein-binding site.
Probe Design and Labeling
- Sequence: Typically a 20-40 bp double-stranded DNA or RNA oligonucleotide.
- Labeling: End-labeling with γ-32P-ATP (for high sensitivity) or with non-radioactive tags (e.g., biotin, fluorophores) is standard.
- Purification: Must be purified post-labeling (e.g., via gel electrophoresis or column purification) to remove unincorporated nucleotides.
Quantitative Data on Probe Characteristics
Table 1: Common Nucleic Acid Probe Labeling Methods
| Method | Typical Specific Activity | Detection Method | Optimal Use-Case | Stability |
|---|---|---|---|---|
| Radioisotopic (³²P) | 10⁸ - 10⁹ cpm/µg | Autoradiography/ Phosphorimaging | High-sensitivity, quantitative assays; competition experiments | ~2 weeks (physical decay) |
| Biotin | N/A | Chemiluminescence (Streptavidin-HRP) | Lab environments avoiding radioactivity; good sensitivity | Months to years |
| Fluorophore (e.g., Cy5, FAM) | N/A | Fluorescence imaging | Multiplexing, real-time kinetics in specialized systems | Months (light-sensitive) |
| Digoxigenin (DIG) | N/A | Chemiluminescence/Colorimetry (Anti-DIG-Ab) | Versatile alternative to biotin; high specificity | Months to years |
Core Component 2: Protein Extract
The source of DNA/RNA-binding proteins, which can be nuclear, cytoplasmic, or whole-cell extracts, or purified/recombinant proteins.
Extract Preparation Protocol
Protocol: Preparation of a HeLa Cell Nuclear Extract for EMSA
- Harvesting: Grow HeLa cells to 80-90% confluence. Wash monolayer with ice-cold PBS.
- Hypotonic Lysis: Scrape cells into a hypotonic buffer (e.g., 10 mM HEPES pH 7.9, 1.5 mM MgCl₂, 10 mM KCl, 0.5 mM DTT, protease inhibitors). Incubate on ice for 15 min. Vortex vigorously for 10 sec.
- Nuclei Pelletation: Centrifuge at 3,300 x g for 15 min at 4°C. The pellet contains nuclei.
- Nuclear Extraction: Resuspend nuclear pellet in a high-salt buffer (e.g., 20 mM HEPES pH 7.9, 25% glycerol, 0.42 M NaCl, 1.5 mM MgCl₂, 0.2 mM EDTA, 0.5 mM DTT, protease inhibitors). Rock gently at 4°C for 30 min.
- Clarification: Centrifuge at 20,000 x g for 30 min at 4°C. Aliquot the supernatant (nuclear extract) and store at -80°C.
- Quantification: Determine protein concentration using the Bradford or BCA assay. Typical yields: 2-5 mg/ml from 10⁷ cells.
Critical Additives for Binding Reactions
- Non-specific Competitors: Poly(dI-dC) or salmon sperm DNA (0.05-0.1 µg/µL) to reduce non-specific probe retention.
- Carrier Protein: BSA (0.1 mg/mL) to stabilize dilute proteins.
- Salts & DTT: KCl/NaCl (50-100 mM) and DTT (0.5-1 mM) to maintain ionic strength and reduce oxidation.
- Detergents: Non-ionic detergents (e.g., NP-40, 0.01-0.1%) can reduce aggregation.
Core Component 3: The Non-Denaturing Gel
The matrix that separates protein-nucleic acid complexes from free probe based on size and charge, without disrupting non-covalent interactions.
Gel Formulation and Electrophoresis Conditions
Protocol: Casting and Running a 6% Non-Denaturing Polyacrylamide Gel
- Gel Composition: Mix 3.0 mL of 30% acrylamide:bis-acrylamide (29:1), 5.0 mL of 5x TBE (or 0.5x TBE for RNA probes), 11.8 mL dH₂O, 150 µL of 10% ammonium persulfate (APS), and 15 µL of TEMED. Pour immediately between glass plates.
- Electrophoresis Buffer: Use 0.5x TBE (for DNA) or 0.5x TBE/1x TBE (for RNA) as the running buffer. Pre-run the gel at 100 V for 30-60 min at 4°C to equilibrate temperature and remove persulfate.
- Sample Loading: Mix binding reaction (see Section 5) with a non-denaturing loading dye (e.g., 10% glycerol, 0.01% xylene cyanol/bromophenol blue). Load immediately.
- Run Conditions: Run at constant voltage (100-150 V) for 1.5-2 hours at 4°C until the dye front migrates appropriately. Maintain temperature to prevent complex dissociation.
Table 2: Non-Denaturing Gel Parameters for EMSA
| Parameter | DNA-Protein EMSA | RNA-Protein EMSA | Notes |
|---|---|---|---|
| Acrylamide % | 4-8% | 4-10% | Lower % for larger complexes. |
| Crosslinker Ratio | 29:1 to 80:1 (Acrylamide:Bis) | 29:1 to 80:1 | Higher bis gives tighter mesh. |
| Gel Buffer | 0.25x - 0.5x TBE or TG | 0.5x TBE or TBE/TAE | Lower ionic strength for sharper bands. |
| Running Buffer | 0.25x - 0.5x TBE or TG | 0.5x TBE or TBE/TAE | Match gel buffer. RNA often uses TBE for RNase inhibition. |
| Running Temperature | 4-10°C | 4-10°C (or room temp) | Critical for complex stability. |
| Voltage Gradient | 6-10 V/cm | 6-10 V/cm | Low voltage minimizes heating. |
Integrated Experimental Protocol: Standard EMSA Workflow
Binding Reaction:
- In a final volume of 10-20 µL, combine:
- 1x Binding Buffer (10 mM HEPES/Tris, pH 7.5-8.0, 50-100 mM KCl/NaCl, 1 mM DTT, 1 mM EDTA, 5-10% Glycerol).
- 0.05-0.1 µg/µL non-specific competitor (poly(dI-dC)).
- 1-10 µg of protein extract (or 1-100 nM purified protein).
- (Optional) Unlabeled competitor DNA/RNA or antibody for supershift.
- Incubate at room temperature or 4°C for 10-20 min.
- Add 10-20 fmol (≈20,000 cpm for ³²P) of labeled probe.
- Incubate for an additional 20-30 min.
- In a final volume of 10-20 µL, combine:
Electrophoresis & Detection:
- Load reaction mix onto pre-run non-denaturing gel.
- Run as per Table 2.
- Transfer gel to blotting membrane (for non-radioactive probes) or dry gel (for ³²P).
- Visualize via appropriate method (Table 1).
Standard EMSA Experimental Workflow from Component Prep to Detection.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for EMSA
| Reagent/Material | Function & Rationale | Example Product/Specification |
|---|---|---|
| Poly(dI-dC) | Non-specific competitor; binds and titrates out non-sequence-specific nucleic acid-binding proteins to reduce background. | High molecular weight, pharmacia grade. |
| Protease Inhibitor Cocktail | Prevents degradation of transcription factors/nucleic acid-binding proteins during extract preparation and binding. | EDTA-free for metal-dependent proteins. |
| RNase Inhibitor (e.g., RNasin) | Essential for RNA EMSA (REMSA); protects RNA probe from degradation. | 40 U/µL. |
| Non-radioactive Labeling Kit | For biotin, DIG, or fluorophore probe labeling; enables safe, stable detection. | e.g., LightShift Chemiluminescent EMSA Kit. |
| High-Binding 0.45 µm Nylon Membrane | For transfer of non-radioactive EMSAs; binds nucleic acid complexes for subsequent detection steps. | Positively charged nylon membrane. |
| Chemiluminescent Substrate (e.g., HRP) | For detection of biotin or DIG-labeled probes after transfer; offers high sensitivity. | Luminol/peroxide-based stable substrate. |
| Gel Drying Apparatus | For drying polyacrylamide gels post-electrophoresis for radioactive detection via phosphorimager. | Slab gel dryer with heating. |
| Cooled Circulator/Electrophoresis Unit | Maintains gel at 4-10°C during run to prevent dissociation of protein-nucleic acid complexes. | Recirculating chiller or cold room setup. |
This whitepaper details the primary applications of techniques used to identify sequence-specific nucleic acid-protein interactions, with a particular focus on the Electrophoretic Mobility Shift Assay (EMSA). This content is framed within a broader research thesis investigating the distinction between "EMSA" and the more generic term "gel retardation assay." While often used synonymously, a key thesis posits that "EMSA" specifically implies the use of a native gel electrophoresis system to detect the complex, whereas "gel retardation assay" can encompass a wider array of separation matrices and conditions. The precision in terminology is critical for experimental design and data interpretation in molecular biology, drug discovery, and functional genomics.
Core Principles and Quantitative Comparison
The fundamental principle involves the separation of a labeled nucleic acid probe from its protein-bound complex using non-denaturing gel electrophoresis. The protein-bound complex migrates more slowly ("shifts" or is "retarded”) than the free probe. Key quantitative parameters defining the assay are summarized below.
Table 1: Core Quantitative Parameters in EMSA/Gel Retardation Assays
| Parameter | Typical Range/Value | Impact on Assay | Notes |
|---|---|---|---|
| Probe Concentration | 0.1-10 nM (labeled) | Below Kd for accurate binding measurement. | Must be in excess for quantitative analysis of protein concentration. |
| Protein Concentration | Varies widely (pM to µM) | Determines fraction of probe bound. | Titration yields apparent dissociation constant (Kd). |
| Poly(dI:dC) Concentration | 0.05-0.5 µg/µL | Competes for non-specific binding. | Critical for reducing background; optimal amount is empirical. |
| Electrophoresis Voltage | 80-150 V | Affects complex stability and resolution. | Higher voltage may cause complex dissociation; typically run at 4-10°C. |
| Gel Percentage (Polyacrylamide) | 4-8% | Resolves complexes of different sizes/shapes. | Lower % for larger complexes (>500 kDa). |
| Electrophoresis Buffer Ionic Strength | 0.25-0.5x TBE or TAE | Maintains complex integrity. | High ionic strength can disrupt weak interactions. |
| Incubation Time (Binding) | 20-30 minutes | Allows equilibrium to be reached. | Longer incubations risk protein/nucleic acid degradation. |
| Detection Sensitivity (Chemiluminescence) | Low femtomole range | Sufficient for most nuclear extracts. | Radioactive (32P) detection can be more sensitive. |
Table 2: Distinguishing EMSA from Broader Gel Retardation Assay (Thesis Context)
| Feature | Electrophoretic Mobility Shift Assay (EMSA) | Gel Retardation Assay (Generic Term) |
|---|---|---|
| Primary Separation Matrix | Native polyacrylamide gel (predominantly). | Can include agarose, composite gels, or other matrices. |
| Typical Application | Protein-DNA/RNA complexes of moderate size (10-500 kDa). | Larger complexes (e.g., nucleoprotein assemblies), very large proteins. |
| Gel State | Non-denaturing, native conditions throughout. | May sometimes use semi-denaturing conditions. |
| Terminology Precision | Specific, technique-defined. | More general, descriptive of the observable result. |
| Thesis Interpretation | Represents a specific, widely adopted methodology. | Describes a broader phenomenon of reduced electrophoretic mobility. |
Detailed Experimental Protocol: Standard EMSA
Objective: To detect and characterize the interaction between a purified transcription factor (e.g., NF-κB) and its cognate DNA sequence.
I. Reagent Preparation
- Probe Labeling: End-label 1-50 pmol of a double-stranded oligonucleotide containing the target sequence with [γ-32P]ATP using T4 Polynucleotide Kinase. Purify using a spin column.
- Binding Buffer (10X Stock): 100 mM Tris-HCl (pH 7.5), 500 mM KCl, 10 mM DTT, 10 mM EDTA, 50% Glycerol. Store at -20°C.
- Polyacrylamide Gel (4%): Mix 3.3 mL 30% acrylamide/bis (29:1), 8.3 mL 1X TBE, 18.4 mL H2O, 200 µL 10% APS, 40 µL TEMED. Pour and allow to polymerize for 30-60 minutes.
- Running Buffer: 0.5X TBE (45 mM Tris-borate, 1 mM EDTA).
II. Binding Reaction
- Assemble a 20 µL reaction on ice:
- 2 µL 10X Binding Buffer
- 1 µL Poly(dI:dC) (1 µg/µL stock)
- 1 µL BSA (10 mg/mL stock)
- X µL Nuclear extract or purified protein (typically 2-10 µg total protein)
- Y µL H2O to volume
- 1 µL 32P-labeled DNA probe (~20 fmol, 50,000 cpm)
- Control Reactions are essential: Include a "probe-only" lane and a "competition" lane with a 100-fold molar excess of unlabeled specific or mutant oligonucleotide.
- Incubate at room temperature (20-25°C) for 20-30 minutes.
III. Electrophoresis and Detection
- Pre-run the 4% native polyacrylamide gel in 0.5X TBE at 100 V for 30-60 minutes in a cold room (4-10°C).
- Load samples (do not add loading dye with SDS or EDTA; use dye without such components, e.g., 6X DNA loading dye without EDTA, in a separate lane).
- Run the gel at 100-150 V constant voltage until the bromophenol blue dye has migrated ~2/3 of the gel length.
- Transfer the gel to blotting paper, dry under vacuum, and expose to a phosphorimager screen overnight. Alternatively, for non-radioactive probes, follow chemiluminescent detection protocols post-electrophoresis.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for EMSA Experiments
| Item | Function | Critical Notes |
|---|---|---|
| Purified Protein or Nuclear Extract | Source of the DNA/RNA-binding protein. | Protein integrity and activity are paramount. Use fresh extracts or aliquots stored at -80°C. |
| 32P-γ-ATP or Chemiluminescent Labeling Kit | Enables sensitive detection of the nucleic acid probe. | Radioactive labeling offers highest sensitivity; chemiluminescent (e.g., biotin/streptavidin-HRP) is safer. |
| Double-Stranded DNA or RNA Probe | Contains the specific sequence of interest for binding. | Must be high-purity, accurately annealed. HPLC or gel purification is recommended. |
| Non-specific Competitor DNA (Poly(dI:dC)) | Binds and sequesters non-sequence-specific nucleic acid-binding proteins. | Reduces background smearing; titration is required for each new protein source. |
| Native Gel Electrophoresis System | Separates protein-nucleic acid complexes from free probe. | Requires a cooling apparatus (cold room or circulator) to maintain complex stability during the run. |
| Specific & Mutant Unlabeled Competitor Oligos | Validates sequence specificity of the observed complex. | A 100-fold excess of specific competitor should abolish the shift; mutant should not. |
| Antibody for Supershift Assay | Identifies a specific protein within a shifted complex. | Causes a further retardation ("supershift") by binding to the protein in the complex. |
Visualizing EMSA Workflow and Data Interpretation
Title: EMSA Experimental Workflow and Controls
Title: Interpretation of EMSA Gel Results and Controls
Step-by-Step EMSA Protocol: From Probe Design to Data Analysis in Modern Research
Within the broader research context of differentiating between the Electrophoretic Mobility Shift Assay (EMSA) and the gel retardation assay—terms often used interchangeably but with nuanced historical and methodological distinctions—probe design and labeling are fundamental. The choice between radioactive and non-radioactive detection methods directly impacts assay sensitivity, safety, cost, and applicability in drug development and mechanistic studies. This guide provides a technical comparison and detailed protocols for these core methodologies.
Core Principles of Probe Design
For both EMSA and gel retardation assays, the probe is a short, double-stranded DNA or RNA oligonucleotide containing the specific protein-binding sequence. Key design considerations include:
- Sequence Specificity: Must match the consensus binding site of the target protein (e.g., transcription factor).
- Length: Typically 20-50 base pairs to ensure sufficient binding specificity while minimizing non-specific interactions.
- Modification Sites: Must include a 5' or 3' end modification (e.g., phosphate group for kinase labeling, amine group for conjugation) or an internal modified base (e.g., biotin-dUTP) depending on the labeling method.
Labeling Methodologies: Technical Comparison
The following table summarizes the quantitative and qualitative data for the primary labeling strategies.
Table 1: Comparison of Probe Labeling and Detection Methods
| Parameter | Radioactive (³²P) | Chemiluminescent (Biotin/Digoxigenin) | Fluorescent (Fluorophore) |
|---|---|---|---|
| Typical Label | [γ-³²P]ATP or [α-³²P]dNTP | Biotin- or Digoxigenin-dUTP | Cy3, Cy5, FAM, TAMRA |
| Detection Limit | 0.1-1 fmol | 1-10 fmol | 10-100 fmol |
| Signal Duration | Short (isotope decay) | Stable (requires activation) | Stable (photo-bleaching) |
| Exposure Time | Minutes to Hours | Seconds to Minutes | Direct Imaging |
| Primary Hazard | Ionizing Radiation | Chemical Hazards | Light Sensitivity |
| Cost per Experiment | Low (reagents) | Medium | High |
| Waste Disposal | Specialized (radioactive) | Standard Chemical | Standard Chemical |
| Multiplexing | No | Possible with stripping | Yes (multiple colors) |
| Quantification | Excellent linear range | Good linear range | Good, but background sensitive |
| Primary Use Case | Maximal sensitivity, traditional assays | High sensitivity, safer labs | Multiplexing, in-gel quantification |
Detailed Experimental Protocols
Protocol 1: Radioactive End-Labeling with [γ-³²P]ATP
Purpose: To label a DNA probe at the 5' end using T4 Polynucleotide Kinase (PNK). Materials: DNA oligonucleotide, [γ-³²P]ATP, T4 PNK, 10X PNK Buffer, Microspin G-25 Column.
- Reaction Setup: In a microcentrifuge tube, combine:
- 1 µL (100 ng) of single-stranded or annealed oligonucleotide
- 1 µL of 10X T4 PNK Buffer
- 3 µL of [γ-³²P]ATP (~3000 Ci/mmol)
- 1 µL of T4 PNK (10 U/µL)
- Nuclease-free water to 10 µL final volume.
- Incubation: Incubate at 37°C for 30 minutes.
- Termination: Heat-inactivate the enzyme at 65°C for 10 minutes.
- Purification: Pass the reaction mixture through a Microspin G-25 column pre-equilibrated with TE buffer to separate labeled probe from unincorporated nucleotides.
- Quantification: Measure radioactivity using a scintillation counter.
Protocol 2: Non-Radioactive Probe Labeling with Biotin-dUTP
Purpose: To generate a biotinylated probe via nick translation or PCR. Materials: DNA template, Biotin-16-dUTP, dNTP mix, Taq DNA Polymerase, primers.
- PCR Labeling Reaction:
- In a PCR tube, set up a standard 50 µL PCR reaction containing:
- 10-100 ng DNA template
- 0.2 mM dATP, dCTP, dGTP
- 0.15 mM dTTP
- 0.05 mM Biotin-16-dUTP
- 0.2 µM each primer
- 1X PCR Buffer
- 1.25 U Taq DNA Polymerase
- In a PCR tube, set up a standard 50 µL PCR reaction containing:
- Amplification: Run appropriate thermal cycling conditions.
- Purification: Purify the PCR product using a PCR purification kit or ethanol precipitation.
- Verification: Run 5% of the product on an agarose gel to confirm size and yield.
Protocol 3: EMSA with Chemiluminescent Detection
Purpose: To perform a gel retardation assay using a biotinylated probe. Materials: Biotinylated probe, nuclear extract, poly(dI-dC), 6% DNA Retardation Gel, 0.5X TBE Buffer, Nylon Membrane, UV Crosslinker, Chemiluminescent Substrate Kit.
- Binding Reaction: Combine:
- 2 µL of 5X Binding Buffer
- 1 µg of poly(dI-dC)
- 5-10 µg of nuclear protein extract
- 20 fmol of biotinylated probe
- Water to 10 µL. Incubate at room temperature for 20 minutes.
- Electrophoresis: Load reaction onto a pre-run 6% non-denaturing polyacrylamide gel in 0.5X TBE. Run at 100 V at 4°C until dye front migrates appropriately.
- Transfer: Electroblot DNA-protein complexes to a positively charged nylon membrane in 0.5X TBE at 380 mA for 30-60 minutes.
- Crosslinking: UV crosslink the DNA to the membrane at 120 mJ/cm².
- Detection: Block membrane, incubate with Streptavidin-HRP conjugate, wash, and incubate with chemiluminescent substrate. Image using a digital imager.
Signaling Pathways & Workflows
Diagram 1: Probe Labeling and EMSA Detection Workflow
Diagram 2: Chemiluminescent Detection Signal Cascade
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions for Probe-Based Assays
| Reagent / Material | Function in Experiment |
|---|---|
| T4 Polynucleotide Kinase (PNK) | Catalyzes the transfer of the terminal phosphate from [γ-³²P]ATP to the 5'-OH end of DNA/RNA. |
| [γ-³²P]ATP | Radioactive donor molecule providing the detectable ³²P label for the probe. |
| Biotin-16-dUTP / Digoxigenin-dUTP | Modified nucleotide incorporated into DNA to provide a hapten for subsequent non-radioactive detection. |
| Fluorophore-labeled dNTPs (e.g., Cy5-dCTP) | Modified nucleotides enabling direct fluorescent labeling of the probe. |
| Poly(dI-dC) | A non-specific competitor DNA used in binding reactions to reduce background from non-specific protein interactions. |
| Non-denaturing Polyacrylamide Gel (4-6%) | Matrix for separating protein-nucleic acid complexes from free probe based on size/shift. |
| Positively Charged Nylon Membrane | Solid support for transferring and immobilizing DNA from gels for chemiluminescent detection. |
| Streptavidin-Horseradish Peroxidase (SA-HRP) | Conjugate that binds biotin with high affinity and catalyzes the chemiluminescent reaction. |
| Enhanced Chemiluminescence (ECL) Substrate | Solution containing luminol and peroxide; oxidized by HRP to produce light. |
| Phosphor Storage Screen & Scanner | For high-sensitivity, quantitative detection of radioactive signals. |
| Fluorescence Gel Imager | Scanner with appropriate lasers and filters to excite and detect fluorescently labeled probes in-gel. |
Within the context of research comparing Electrophoretic Mobility Shift Assays (EMSAs) with classical gel retardation assays, the integrity of the protein sample is paramount. The reliability of these techniques, which detect protein-nucleic acid interactions, hinges on the quality and specificity of the prepared protein. This technical guide details three core preparation strategies—nuclear extracts, recombinant proteins, and whole-cell lysates—each serving distinct experimental needs in transcription factor and DNA-binding protein research.
Nuclear Extracts
Nuclear extracts are essential for studying DNA-binding proteins, especially transcription factors that translocate to the nucleus. They provide an enriched source of nuclear proteins while reducing cytoplasmic contamination.
Detailed Protocol: Hypotonic Lysis with High-Salt Extraction
Principle: Swell cells in a hypotonic buffer, lyse the plasma membrane, isolate nuclei via centrifugation, and extract nuclear proteins with a high-salt buffer.
Reagents:
- Hypotonic Buffer A: 10 mM HEPES (pH 7.9), 1.5 mM MgCl₂, 10 mM KCl, 0.5 mM DTT, 0.2 mM PMSF, protease inhibitor cocktail.
- High-Salt Extraction Buffer C: 20 mM HEPES (pH 7.9), 25% (v/v) glycerol, 0.42 M NaCl, 1.5 mM MgCl₂, 0.2 mM EDTA, 0.5 mM DTT, 0.2 mM PMSF, protease inhibitors.
Procedure:
- Harvest ~10⁷ cells by centrifugation (500 x g, 5 min, 4°C). Wash with PBS.
- Resuspend pellet in 5x pellet volume of ice-cold Buffer A. Incubate on ice for 15 min.
- Add Nonidet P-40 (NP-40) to 0.5% final concentration. Vortex vigorously for 10 sec.
- Centrifuge immediately (16,000 x g, 30 sec, 4°C). The supernatant is the cytoplasmic fraction.
- Wash the nuclear pellet gently with Buffer A (without NP-40). Centrifuge again.
- Resuspend nuclei in 0.5-1x original pellet volume of Buffer C. Rock gently at 4°C for 30 min.
- Centrifuge (16,000 x g, 10 min, 4°C). Aliquot and snap-freeze the supernatant (nuclear extract) at -80°C.
Workflow Diagram
Recombinant Proteins
Recombinant proteins offer a pure, defined source of protein, critical for validating specific interactions in EMSA and controlling for confounding cellular factors.
Detailed Protocol: Affinity Purification of His-Tagged Protein fromE. coli
Principle: Express protein with a polyhistidine (His) tag in E. coli, lyse cells, and purify via immobilized metal affinity chromatography (IMAC).
Reagents:
- Lysis Buffer: 50 mM Tris-HCl (pH 8.0), 300 mM NaCl, 10 mM imidazole, 1 mg/mL lysozyme, protease inhibitors, 1 mM PMSF.
- Wash Buffer: 50 mM Tris-HCl (pH 8.0), 300 mM NaCl, 25 mM imidazole.
- Elution Buffer: 50 mM Tris-HCl (pH 8.0), 300 mM NaCl, 250 mM imidazole.
- Ni-NTA Agarose Resin.
Procedure:
- Induce expression in E. coli culture with IPTG. Harvest by centrifugation.
- Resuspend pellet in Lysis Buffer. Incubate on ice for 30 min, then sonicate.
- Clarify lysate by centrifugation (20,000 x g, 30 min, 4°C).
- Incubate supernatant with pre-equilibrated Ni-NTA resin for 1 hr at 4°C with gentle mixing.
- Load resin into a column. Wash with 10 column volumes of Wash Buffer.
- Elute protein with 3-5 column volumes of Elution Buffer.
- Dialyze eluted protein into storage buffer (e.g., 20 mM HEPES pH 7.9, 100 mM KCl, 0.2 mM EDTA, 20% glycerol, 1 mM DTT) to remove imidazole.
Whole-Cell Lysates
Whole-cell lysates provide a global view of a cell's protein content, useful for studying proteins distributed across cellular compartments or when nuclear enrichment is not required.
Detailed Protocol: RIPA Buffer Lysis
Principle: Use a Radioimmunoprecipitation Assay (RIPA) buffer containing detergents and salts to solubilize both membrane and cytoplasmic proteins.
Reagents:
- RIPA Buffer: 50 mM Tris (pH 8.0), 150 mM NaCl, 1% NP-40, 0.5% sodium deoxycholate, 0.1% SDS. Add DTT and protease inhibitors fresh.
Procedure:
- Harvest cells (~10⁶-10⁷) and wash with PBS.
- Lyse cells in 100-500 µL of ice-cold RIPA buffer per 10⁶ cells. Incubate on ice for 15-30 min with occasional vortexing.
- Clarify the lysate by centrifugation (16,000 x g, 15 min, 4°C).
- Transfer the supernatant to a new tube. Determine protein concentration via Bradford or BCA assay. Aliquot and store at -80°C.
Comparative Data & Applications
Table 1: Comparison of Protein Sample Types for EMSA/Gel Retardation Studies
| Feature | Nuclear Extract | Recombinant Protein | Whole-Cell Lysate |
|---|---|---|---|
| Primary Use | Study of endogenous nuclear proteins (e.g., transcription factors) | Study of specific, purified DNA-protein interactions | Global analysis of DNA-binding activity; proteins from all compartments |
| Key Advantage | Physiological context & post-translational modifications | High purity, no confounding factors, high yield | Simplicity, speed, preserves some protein complexes |
| Major Limitation | Complexity, risk of proteolysis, cytoplasmic contamination | Lack of native modification & cellular context | High background, dilution of nuclear factors |
| Typical Yield | 0.5-2 mg from 10⁷ HeLa cells | 1-10 mg per liter E. coli culture | 1-5 mg from 10⁷ adherent cells |
| EMSA Suitability | Excellent for endogenous complexes | Ideal for probe validation & competition assays | Suitable when target is abundant or exclusively nuclear |
Table 2: Selection Guide Based on Research Question
| Research Question | Recommended Sample | Rationale |
|---|---|---|
| Does transcription factor X bind to promoter Y in liver cells? | Nuclear extract from liver tissue/cells | Captures endogenous, post-translationally modified TF in its native compartment. |
| What is the precise DNA sequence motif for protein Z? | Purified recombinant protein Z | Eliminates interference from other DNA-binding proteins, allowing clear mapping. |
| Does drug treatment alter the DNA-binding activity of factor A? | Paired nuclear extracts (control vs. treated) | Allows detection of changes in activity due to signaling events or localization. |
| Initial screening for DNA-binding activity of an uncharacterized gene product. | Whole-cell lysate from overexpression system | Fastest way to check for binding before undertaking complex purification. |
The Scientist's Toolkit
Table 3: Essential Reagent Solutions for Protein Sample Preparation
| Reagent | Function | Key Consideration |
|---|---|---|
| Protease Inhibitor Cocktail (e.g., EDTA-free) | Prevents protein degradation by serine, cysteine, metallo-proteases, etc. | Must be added fresh to lysis buffers. EDTA-free versions are essential for metal-dependent processes. |
| PMSF (Phenylmethylsulfonyl fluoride) | Serine protease inhibitor (e.g., trypsin, chymotrypsin). | Unstable in aqueous solution; add immediately before use. Toxic. |
| DTT (Dithiothreitol) | Reducing agent; maintains cysteine residues in reduced state. | Critical for preserving activity of many DNA-binding proteins. Add fresh. |
| Glycerol (20-50%) | Stabilizes protein structure, prevents aggregation, allows storage at -20°C/-80°C. | Standard component of storage/lysis buffers for long-term activity retention. |
| NP-40 / IGEPAL CA-630 | Non-ionic detergent for cell membrane lysis. Mild, preserves protein-protein interactions. | Concentration is critical (0.1-1%). Used in nuclear and whole-cell lysis. |
| Imidazole | Competes with His-tag for binding to Ni²⁺ resin; used for washing and elution. | Low (10-30 mM) for washing, high (150-500 mM) for elution in recombinant protein purification. |
| High-Salt Buffer (NaCl/KCl >400 mM) | Disrupts ionic interactions to extract proteins from nucleic acids (nuclei) or columns. | Salt concentration must be optimized to extract target without co-extracting contaminants. |
| HALT Phosphatase Inhibitor Cocktail | Preserves phosphorylation states, crucial for studying regulated transcription factors. | Added to lysis buffers when studying signaling-responsive DNA-binding activity. |
Contextualizing Preparation for EMSA/Gel Retardation Assays
The choice of protein sample directly influences the interpretation of EMSA results. Nuclear extracts are indispensable for studying physiological complexes in diseases like cancer, where transcription factor activity is aberrantly regulated. Recombinant proteins serve as the gold standard for defining binding specificity, a core requirement when arguing that a shifted band represents a bona fide interaction. Whole-cell lysates, while less specific, can be valuable in high-throughput screens. In the ongoing methodological discourse, "EMSA" often implies a more sophisticated setup (e.g., using supershift antibodies or cold competition), but its foundation is the "gel retardation assay," for which any of these samples can be used. The rigor of the sample preparation dictates the validity of the observed retardation.
Pathway: From Sample to EMSA Result
The electrophoretic mobility shift assay (EMSA) and the historically synonymous term "gel retardation assay" represent a cornerstone technique for studying nucleic acid-protein interactions. Within the broader thesis of differentiating EMSA from gel retardation assay research, it is critical to recognize that "gel retardation assay" is the original descriptive name, while "EMSA" is the modern, more precise terminology for the same core methodology. The distinction lies not in the fundamental principle—both monitor the reduction in electrophoretic mobility of a nucleic acid probe upon protein binding—but in the evolution of the technique's sophistication. Contemporary EMSA research focuses on rigorous optimization of the binding reaction to ensure specificity, affinity, and quantitative accuracy, moving beyond simple qualitative retardation observations. This guide details the critical parameters governing this central binding reaction.
Core Components of the Binding Reaction Optimization
Buffer Composition
The binding buffer establishes the ionic and pH environment crucial for productive interaction. Key components include:
- pH Buffer (e.g., HEPES, Tris): Maintains a stable pH, typically between 7.0 and 8.5.
- Salts (KCl, NaCl): Modulate ionic strength; low ionic strength can promote non-specific binding.
- Divalent Cations (Mg²⁺, Zn²⁺): Often required for the structural integrity of protein or DNA motifs.
- Reducing Agents (DTT, β-mercaptoethanol): Maintain cysteines in reduced state, preserving protein function.
- Non-ionic Detergents (NP-40, Tween-20): Reduce non-specific adsorption (e.g., to tubes).
- Carrier Proteins (BSA, Ficoll, Polyvinyl alcohol): Reduce non-specific binding by occupying low-affinity sites.
Table 1: Common Binding Buffer Components and Their Functions
| Component | Typical Concentration | Primary Function | Optimization Consideration |
|---|---|---|---|
| HEPES, pH 7.9 | 10-20 mM | pH stabilization | Preferred over Tris for minimal temperature sensitivity. |
| KCl | 50-150 mM | Controls ionic strength | High conc. (>200 mM) may disrupt specific complexes. |
| MgCl₂ | 1-10 mM | Cofactor for DNA-binding | Essential for many transcription factors; omit for non-requiring proteins. |
| DTT | 0.5-1.0 mM | Reducing agent | Fresh preparation is critical; prevents oxidation of protein thiols. |
| NP-40 / Tween-20 | 0.01-0.1% | Non-specific binding reduction | Minimizes protein adhesion to plastics. |
| BSA / Ficoll-400 | 0.1-1.0 mg/mL | Carrier / Stabilizer | Competes for non-specific sites; stabilizes dilute proteins. |
Competitors for Specificity
Competitor nucleic acids are vital for distinguishing specific from non-specific complexes.
- Non-specific Competitors: Poly(dI-dC) • poly(dI-dC) is most common. It sequesters proteins with general affinity for the DNA backbone.
- Specific Competitors: An unlabeled oligonucleotide identical to the probe. Used in "cold competition" experiments to confirm binding specificity.
Table 2: Competitor Nucleic Acids in EMSA
| Competitor Type | Example | Typical Amount per Reaction | Purpose & Mechanism |
|---|---|---|---|
| Non-specific | Poly(dI-dC) • Poly(dI-dC) | 0.05-0.5 µg | Blocks non-specific electrostatic interactions between protein and probe DNA backbone. |
| Non-specific | Sheared Salmon Sperm DNA | 0.1-1.0 µg | Alternative to poly(dI-dC); useful for some protein families. |
| Specific (Cold) | Unlabeled identical probe | 5-100x molar excess over labeled probe | Competes for binding to the specific protein; confirms sequence specificity. |
| Mutant Specific | Unlabeled probe with mutated binding site | 5-100x molar excess | Serves as a negative control; should not effectively compete for specific binding. |
Incubation Conditions
- Temperature and Time: Binding reactions are typically incubated at 20-30°C for 15-30 minutes. Ice-cold incubation (0-4°C) for longer periods (up to 45 min) can be used for less stable complexes.
- Order of Addition: A standard protocol is to add buffer, competitor, protein extract, and finally the labeled probe. Pre-incubating protein with competitor for 5-10 minutes before probe addition can enhance specificity.
Detailed Experimental Protocol: A Standard EMSA Binding Reaction
Objective: To form a specific protein-nucleic acid complex for electrophoretic analysis.
Materials:
- Purified protein or nuclear extract.
- ³²P-end-labeled or fluorescently-labeled DNA/RNA probe.
- 2X or 5X concentrated binding buffer stock.
- Competitor DNA stock solutions (poly(dI-dC) and unlabeled specific probe).
- Nuclease-free water.
Procedure:
- Prepare Master Mix (for n reactions + 10% extra): In a nuclease-free microcentrifuge tube, combine the following per reaction:
- Nuclease-free water (to bring to final volume).
- 2µL of 5X binding buffer (final: 1X).
- 1µL of 1µg/µL poly(dI-dC) (final: 1 µg).
- 0.5µL of 100 mM DTT (final: 5 mM).
- Optional: 0.5µL of 20% Ficoll-400 (final: 2%).
- Aliquot: Dispense the appropriate volume of Master Mix into individual reaction tubes.
- Add Competitor (for competition assays): Add the desired molar excess of unlabeled specific or mutant probe to the respective tubes. Include a "no competitor" control.
- Add Protein: Add 2-10 µg of nuclear extract or 10-200 ng of purified protein to each tube. Mix gently by pipetting. Pre-incubate at room temperature for 10 minutes.
- Initiate Binding: Add 1µL of labeled probe (10-50 fmol, ~20,000 cpm for radioactive) to each tube. Mix gently. Do not vortex.
- Incubate: Incubate the reaction at room temperature (20-25°C) for 20-30 minutes.
- Load Gel: After incubation, add 1-2µL of 10X non-denaturing loading dye (without SDS or bromophenol blue that can disrupt complexes). Load immediately onto a pre-run non-denaturing polyacrylamide gel.
Visualizations
Title: EMSA Binding Reaction Workflow
Title: Optimization & Validation Pathway for EMSA
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents for EMSA Binding Reactions
| Reagent | Function & Role in Optimization | Example Vendor/Product Notes |
|---|---|---|
| High-Purity Buffers (HEPES, Tris) | Maintains precise reaction pH; affects protein conformation and binding affinity. | Molecular biology grade, nuclease-free stocks. |
| Nuclease-Free Water | Prevents degradation of nucleic acid probes and competitors. | Certified DEPC-treated or ultrapure filtered. |
| Poly(dI-dC) • Poly(dI-dC) | Gold-standard non-specific competitor for DNA-protein EMSAs; length and concentration are critical variables. | Pharmacia or equivalent high-purity preparation. |
| DTT or β-Mercaptoethanol | Reducing agent preserving protein activity; fresh aliquots required. | >0.5M stock solutions stored at -20°C. |
| Protease Inhibitor Cocktails | Essential when using cell/ tissue extracts; prevents protein degradation during incubation. | EDTA-free cocktails if divalent cations are needed. |
| Carrier Proteins (BSA, Ficoll) | Stabilizes dilute proteins and reduces surface adhesion. | Acetylated BSA is often preferred to minimize enzymatic activity. |
| Radioisotope or Chemiluminescent Labeling Kits | For probe labeling; sensitivity dictates probe amount needed in binding reaction. | [γ-³²P] ATP & T4 PNK, or biotin/fluorescent labeling kits. |
| Non-denaturing Loading Dye | Adds density for gel loading without disrupting weak non-covalent complexes. | Contains glycerol or Ficoll; lacks SDS and denaturants. |
This whitepaper provides an in-depth technical guide to non-denaturing (native) gel electrophoresis, focusing on the critical choice between polyacrylamide and agarose matrices and their associated buffer systems. The content is framed within the context of research on nucleic acid-protein interactions, specifically addressing the methodologies underpinning the Electrophoretic Mobility Shift Assay (EMSA), also known as the gel retardation assay. While often used interchangeably, EMSA typically implies a specific application for detecting protein binding to a nucleic acid probe, whereas "gel retardation assay" can be a broader term. The core principle—separation of complexes based on mobility shifts in a native gel—is common to both. This guide details the technical considerations for optimizing these assays.
Core Principles and Matrix Selection
The fundamental principle is the electrophoretic separation of biomolecular complexes under conditions that preserve their native, folded structure and non-covalent interactions. The choice of gel matrix is paramount and is dictated by the size range of the analytes.
Polyacrylamide Gels are formed by the polymerization of acrylamide and bis-acrylamide, creating a tunable, cross-linked porous mesh. They are ideal for separating smaller complexes (typically <500 kDa) with high resolution. This makes them the standard for classic EMSA studies of transcription factors bound to short DNA or RNA oligonucleotides (20-50 bp).
Agarose Gels are formed by polymerizing agarose polysaccharide chains, resulting in larger pores. They are suitable for separating larger complexes (>500 kDa), such as multi-protein complexes on long DNA fragments, nucleoprotein complexes, or very large protein oligomers.
Quantitative Comparison of Gel Matrices
Table 1: Comparative Properties of Polyacrylamide and Agarose Gels for Native Electrophoresis
| Property | Polyacrylamide Gel | Agarose Gel |
|---|---|---|
| Typical Concentration | 4-10% (w/v) | 0.5-2% (w/v) |
| Effective Separation Range | ~5 kDa to 500 kDa | ~50 kDa to several MDa |
| Pore Size Control | Precise, via %T and %C | Limited, primarily by agarose % |
| Resolution | High | Moderate |
| Sample Capacity | Low | High |
| Typical Thickness | 0.5-1.5 mm | 3-10 mm |
| Standard Format | Vertical system | Horizontal (submerged) system |
| Primary Application in EMSA | Small protein-nucleic acid complexes | Large nucleoprotein complexes (e.g., chromatin) |
Buffer Systems for Native Electrophoresis
The buffer system serves two key functions: maintaining pH during electrophoresis and preserving native interactions. Two main configurations exist: continuous and discontinuous (disc) systems.
Continuous Buffer Systems: The same buffer is used in the tank and gel. Simpler to set up but offers less sharpness in band definition. Common for agarose native gels and many polyacrylamide EMSAs.
- Tris-Glycine (pH 8.3-8.8): Common, wide-range buffer.
- Tris-Borate-EDTA (TBE, pH ~8.3): Provides buffering capacity and chelates divalent cations.
- Tris-Acetate-EDTA (TAE, pH ~8.3): Lower buffering capacity than TBE.
Discontinuous (Disc) Buffer Systems: Employ different buffers in the gel (stacking gel buffer) and tank (running buffer). The stacking gel concentrates samples into a sharp zone before entering the resolving gel, yielding higher resolution. The native Tris-Glycine system is a classic example.
Key Buffer Components and Considerations:
- pH: Typically 8.0-8.8 to maintain protein stability and charge.
- Ionic Strength: Low to moderate (e.g., 5-50 mM) to minimize heat generation. Binding reactions are often performed in a separate, higher-salt binding buffer.
- Divalent Cations: Mg²⁺ or Zn²⁺ may be included if essential for complex stability.
- Co-factors/Reducing Agents: DTT or β-mercaptoethanol can be added to prevent oxidation.
- Glycerol: Often included in loading buffer (5-10% v/v) to increase sample density.
Table 2: Common Native Electrophoresis Buffer Systems
| Buffer System | Type | Typical Use | Critical Notes |
|---|---|---|---|
| 0.5X TBE | Continuous | Native PAGE & Agarose | Common for DNA-protein EMSA; EDTA may affect metal-dependent binding. |
| Tris-Glycine (pH 8.8) | Continuous or Disc | Native PAGE | Versatile; compatible with many proteins. Disc system offers superior stacking. |
| TAE | Continuous | Native Agarose | For very large complexes; lower buffering capacity requires recirculation for long runs. |
| HEPES-Based | Continuous | Specialized EMSA | Mimics physiological pH (7.0-7.5) more closely than Tris-based buffers. |
Experimental Protocol: Standard EMSA/Gel Retardation Assay
Materials & Reagents
- Purified Protein: Recombinant or nuclear extract.
- Nucleic Acid Probe: 20-50 bp DNA/RNA, end-labeled with ³²P, biotin, or fluorophore.
- Binding Buffer: 10-20 mM HEPES/Tris (pH 7.5-8.0), 50-100 mM KCl/NaCl, 1-5 mM MgCl₂, 0.5-1 mM DTT, 0.1-0.2 mg/mL BSA, 5-10% glycerol, 0.1% NP-40.
- Non-specific Competitor DNA: Poly(dI-dC) or sheared salmon sperm DNA.
- Specific Competitor DNA: Unlabeled identical probe.
- Polyacrylamide Gel (4-6%): Pre-cast in 0.5X TBE or Tris-Glycine.
- Electrophoresis Unit & Power Supply.
- Detection System: Phosphorimager (radioactive), CCD camera (chemiluminescent), or fluorescence scanner.
Methodology
- Probe Preparation: Label oligonucleotide probe at 5' or 3' end. Purify using spin column.
- Binding Reaction:
- Assemble 10-20 μL reaction on ice: Binding Buffer, 1-10 μg nuclear extract or 10-100 ng purified protein, 0.1-1 μg poly(dI-dC), 1-10 fmol labeled probe.
- Include controls: probe alone, probe + protein, probe + protein + 100x molar excess specific competitor.
- Incubate 20-30 minutes at room temperature.
- Gel Loading & Electrophoresis:
- Pre-run gel in cold room (4-10°C) at 100V for 30-60 min in appropriate running buffer.
- Add non-ionic loading dye (e.g., 10% glycerol, 0.01% bromophenol blue) to reactions. Do not use SDS or heat.
- Load samples immediately. Run at constant voltage (100-150V for mini-gels) until dye migrates 2/3 of the gel. Maintain cold temperature to prevent complex dissociation.
- Detection:
- For radioactive probes: Dry gel and expose to phosphor screen.
- For chemiluminescent/biotin probes: Transfer to membrane, crosslink, and develop with streptavidin-HRP.
- For fluorescent probes: Scan gel directly.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Non-Denaturing EMSA/Retardation Assays
| Reagent/Material | Function | Example/Critical Specification |
|---|---|---|
| High-Purity Acrylamide/Bis Mix (29:1 or 37.5:1) | Forms the polyacrylamide gel matrix. | RNase/DNase-free, electrophoresis grade. |
| Non-Denaturing Loading Buffer | Increases sample density for well loading without disrupting complexes. | Contains glycerol (10-30%) and inert tracking dyes (bromophenol blue/xylene cyanol). |
| Poly(dI-dC)·Poly(dI-dC) | Non-specific competitor DNA to reduce non-specific protein-nucleic acid binding. | Highly polymerized, sonicated. |
| Protease & Phosphatase Inhibitor Cocktails | Preserve protein integrity and phosphorylation state in crude extracts. | Added to extraction and binding buffers. |
| Chemiluminescent Nucleic Acid Detection Kit | Non-radioactive detection of biotin- or DIG-labeled probes. | Includes streptavidin-HRP conjugate and enhanced chemiluminescence substrate. |
| Fluorescently-Labeled Nucleotides (Cy3, Cy5, FAM) | For safe, direct detection of probes in gels. | Used in place of ³²P for probe labeling. |
| Pre-cast Native PAGE Gels | Provide consistency and save time in gel preparation. | Available in various % and well formats, often with buffer kits. |
| Mobility Shift Assay (EMSA) Buffer Kits | Optimized, ready-to-use buffers for binding and electrophoresis. | Ensure reproducibility and often include positive controls. |
Visualizing the Workflow and Logic
Title: EMSA Experimental Control Workflow
Title: Native Gel Matrix Selection Guide
This technical guide details the core methodologies of autoradiography, imaging, and densitometric analysis, with a specific focus on their application in the differentiation and validation of Electrophoretic Mobility Shift Assay (EMSA) and gel retardation assay data. These techniques are fundamental for the quantitative analysis of nucleic acid-protein interactions, crucial for research in gene regulation and drug discovery targeting transcription factors.
Within the context of EMSA/gel retardation assay research, the core challenge is not just to separate protein-nucleic acid complexes but to precisely detect, visualize, and quantify them. While the terms are often used interchangeably, a technical distinction exists: "gel retardation assay" describes the principle of separation, while "EMSA" explicitly incorporates the detection methodology. This guide focuses on the critical detection and quantification phase, which validates the observed shift and provides quantitative data on binding affinity, stoichiometry, and kinetics.
Section 1: Autoradiography - The Traditional Gold Standard
Autoradiography uses radioactive isotopes (e.g., ³²P, ³⁵S) to label nucleic acid probes, providing high sensitivity for detecting shifted complexes in EMSAs.
Key Research Reagent Solutions
| Item | Function in EMSA/Retardation Assay |
|---|---|
| [γ-³²P]ATP or [α-³²P]dNTP | Radioactive label incorporated into the DNA or RNA probe via kinase or fill-in reactions. |
| T4 Polynucleotide Kinase | Catalyzes the transfer of the γ-phosphate from ATP to the 5'-end of oligonucleotides. |
| Poly(dI•dC) or Non-Specific DNA | Non-specific competitor to reduce background from non-specific protein binding. |
| Phosphor Storage Screen | A reusable screen that stores latent energy from beta particles emitted by the radioisotope. |
| X-ray Film | Traditional silver halide film for direct exposure autoradiography. |
| Autoradiography Cassette | Light-tight, rigid casing to hold the gel/membrane and film/screen in intimate contact. |
Detailed Protocol: EMSA Probe Labeling and Autoradiography
- End-Labeling Reaction: Combine 1-10 pmol of purified oligonucleotide, 2 µL of 10X T4 PNK buffer, 50 µCi of [γ-³²P]ATP, 10 U of T4 Polynucleotide Kinase, and nuclease-free water to 20 µL. Incubate at 37°C for 30-60 minutes.
- Purification: Remove unincorporated nucleotides using a spin column (e.g., Sephadex G-25) or ethanol precipitation.
- EMSA & Electrophoresis: Perform standard binding reaction and run on a non-denaturing polyacrylamide gel (typically 4-8%).
- Gel Transfer: Carefully transfer the gel onto filter paper and dry under vacuum at 80°C for 1-2 hours.
- Direct Autoradiography: In a darkroom, place the dried gel in a cassette with X-ray film. Expose at -80°C for several hours to days. Develop the film using an automated processor.
- Indirect Autoradiography (Phosphor Imaging): Place the dried gel in direct contact with a Phosphor Storage Screen. Exposure time is typically 1/10th of that required for film.
- Scanning: Scan the exposed Phosphor Screen in a Phosphor Imager or Laser Scanner (e.g., Typhoon, Storm). The laser excites the trapped energy, releasing light proportional to the radioactivity, which is captured by a photomultiplier tube.
Section 2: Modern Imaging Alternatives
Non-radioactive detection methods have become prevalent, offering safety and convenience.
| Method | Label | Detection Principle | Relative Sensitivity |
|---|---|---|---|
| Chemiluminescence | Biotin, Digoxigenin | Enzyme (HRP/AP)-catalyzed light emission on film/CCD. | High (comparable to ³²P) |
| Fluorescence | Cy3, Cy5, FAM | Direct excitation/emission by laser scanner. | Moderate-High |
| Colorimetric | Biotin, Digoxigenin | Enzyme (HRP/AP) produces insoluble colored precipitate. | Low-Moderate |
Detailed Protocol: Chemiluminescent Detection for EMSA
- Electroblotting: After electrophoresis, transfer complexes from the gel to a positively charged nylon membrane via wet or semi-dry transfer.
- Crosslinking: UV-crosslink nucleic acids to the membrane (120 mJ/cm²).
- Blocking: Incubate membrane in 5% non-fat dry milk/TBST for 1 hour.
- Conjugate Binding: Incubate with Streptavidin-Horseradish Peroxidase (HRP) (for biotin) or Anti-Digoxigenin-AP (for DIG) in blocking buffer for 30-60 minutes.
- Washing: Wash membrane 3 x 5 minutes with TBST.
- Substrate Incubation: Incubate with enhanced chemiluminescent (ECL) substrate for HRP or CSPD for AP as per manufacturer's instructions.
- Imaging: Capture the chemiluminescent signal using a CCD-based imaging system (e.g., ChemiDoc), optimizing exposure time (seconds to minutes).
Section 3: Densitometric Analysis for Quantification
Densitometry converts signal intensity from autoradiographs or digital images into quantitative data for determining dissociation constants (Kd) or binding kinetics.
Quantitative Data Analysis Workflow
Table 1: Key Quantitative Outputs from EMSA Densitometry
| Parameter | Description | Formula/Notes |
|---|---|---|
| Fraction Bound (θ) | Proportion of total probe bound to protein. | θ = Intensity(Complex) / [Intensity(Complex) + Intensity(Free Probe)] |
| Dissociation Constant (Kd) | Measure of binding affinity. Lower Kd = higher affinity. | Derived from non-linear regression of θ vs. [Protein] using the Hill equation or specific binding models. |
| Hill Coefficient (nH) | Indicates cooperativity in binding. nH>1 = positive cooperativity. | Parameter from the Hill equation fit. |
| IC50 (Competition EMSA) | Concentration of unlabeled competitor that displaces 50% of labeled probe. | Determined from dose-response curve of competitor vs. fraction bound. |
Detailed Protocol: Densitometric Analysis Using ImageJ/Fiji
- Image Import: Open a 16-bit TIFF image from the phosphor imager or CCD system.
- Background Subtraction: Apply "Rolling Ball" or "Subtract Background" function.
- Lane Profiling: Define rectangular regions of interest (ROIs) for each lane and the background. Use the "Plot Lanes" or "Gel Analyzer" tool.
- Peak Integration: Manually or automatically select peaks corresponding to free probe and shifted complexes. Record the integrated intensity (volume) for each.
- Data Normalization: Correct for background intensity from an adjacent area with no signal.
- Curve Fitting: Export intensity data to graphing software (e.g., GraphPad Prism). Calculate Fraction Bound (θ) for each protein concentration. Fit the θ vs. [Protein] data to a one-site specific binding (hyperbola) or Hill equation model to derive Kd and nH.
Visualizing the Workflow and Data Logic
Diagram 1: EMSA Detection and Quantification Workflow (97 chars)
Diagram 2: Densitometric Analysis Data Pipeline (82 chars)
The precise detection and quantification steps outlined here are what transform a simple gel retardation observation into the quantitatively rigorous EMSA. The choice between high-sensitivity autoradiography and safer, modern imaging depends on the required sensitivity, available infrastructure, and regulatory environment. Subsequent densitometric analysis provides the critical numerical data to distinguish specific from non-specific binding and to calculate definitive thermodynamic and kinetic parameters, thereby solidifying conclusions within the broader thesis of nucleic acid-protein interaction studies.
Within the framework of research distinguishing the Electrophoretic Mobility Shift Assay (EMSA) from the broader category of gel retardation assays, a focus on advanced applications highlights the evolution from simple detection of protein-nucleic acid interactions to sophisticated, quantitative, and high-throughput analyses. This guide details the core advanced methodologies that leverage the foundational principle of mobility shift.
Supershift Assays: Identifying Complex Components
A supershift assay is an extension of the standard EMSA used to identify specific proteins within a protein-DNA or protein-RNA complex. An antibody against a suspected protein component is added to the binding reaction. If the antibody binds to the protein within the complex, it creates an even larger "supershifted" complex with a further reduced mobility on the gel.
Experimental Protocol: Supershift Assay
- Prepare Binding Reactions: Set up standard EMSA binding reactions containing your labeled probe and nuclear extract or purified protein(s).
- Antibody Addition: To the experimental reaction, add 1-2 µg of a specific antibody (e.g., anti-p53, anti-NF-κB p65). Include control reactions with: a) no antibody, b) an isotype control/non-specific antibody.
- Pre-incubation: Incubate the reaction mixture (without probe) with the antibody for 20-30 minutes on ice or at room temperature to allow antibody-protein binding.
- Probe Addition & Final Incubation: Add the labeled nucleic acid probe and incubate for an additional 20-30 minutes.
- Electrophoresis: Load the reactions onto a native polyacrylamide gel and run as per standard EMSA protocol.
- Detection: Visualize the complexes. A successful supershift is indicated by a further retardation (higher in the gel) of a portion or all of the original protein-probe complex band.
Diagram: Supershift Assay Workflow
Competition Assays: Measuring Binding Specificity and Affinity
Competition assays quantitatively assess the specificity and relative affinity of a protein for its target nucleic acid sequence. An unlabeled competitor oligonucleotide is included in the binding reaction to compete with the labeled probe for protein binding.
Experimental Protocol: Competition Assay
- Design Competitors:
- Specific (Cold) Competitor: Identical in sequence to the labeled probe.
- Mutant (Non-specific) Competitor: Contains mutations in the known protein-binding site.
- Non-related Competitor: An unrelated sequence.
- Set Up Reactions: Prepare a series of standard binding reactions with a constant amount of protein and labeled probe.
- Titrate Competitor: Add increasing molar excesses (e.g., 5x, 10x, 50x, 100x) of each unlabeled competitor to separate reaction tubes. Include a no-competitor control.
- Incubation & Electrophoresis: Incubate and run on a native gel as per standard protocol.
- Quantification: Use densitometry to measure the intensity of the shifted band. Plot % bound probe vs. competitor molar excess.
Key Quantitative Data from Competition Assays Table 1: Interpretation of Competition Assay Results
| Competitor Type | Expected Result (Band Intensity) | Interpretation |
|---|---|---|
| Specific (Cold) | Decreases with increasing excess | Confirms sequence-specific binding. The excess required indicates relative affinity. |
| Mutant Binding Site | Little to no decrease | Confirms specificity for the exact binding sequence. |
| Non-related Sequence | No change | Further confirms binding specificity, ruling out non-sequence-specific interactions. |
High-Throughput Variations: Moving Beyond the Gel
Modern adaptations transform EMSA into a high-throughput, quantitative platform suitable for drug discovery and large-scale screening.
1. Fluorescence Polarization (FP-EMSA): Uses a fluorescently labeled probe. When bound to a large protein, the fluorescent probe rotates slowly, resulting in high polarization. Free probe rotates quickly, yielding low polarization. Binding is measured in real-time in microplate format.
- Protocol: Titrate protein into a solution containing a low concentration of fluorescent probe in a 384-well plate. Measure fluorescence polarization after a short incubation. Fit data to determine Kd.
2. Microfluidic Capillary Electrophoresis: Automates the separation and detection of complexes, offering superior resolution, quantitation, and speed over slab gels.
- Protocol: Binding reactions are loaded into a capillary. An applied voltage separates free probe from complex. Detection is via laser-induced fluorescence of the labeled probe.
3. EMSA-In-Cell (ICeEMSAs): Adapts the principle to measure protein-nucleic acid interactions directly in crude cellular lysates without purification, preserving native post-translational modifications.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Advanced EMSA Applications
| Item | Function & Rationale |
|---|---|
| High-Affinity, Sequence-Specific Antibodies | For supershift assays; must recognize the native epitope of the protein in the complex. |
| Biotin- or Fluorescently-Labeled Nucleotides | For non-radioactive probe labeling, compatible with chemiluminescent detection or FP-EMSA. |
| Chemically Synthesized Competitor Oligonucleotides | Unlabeled, specific, and mutant sequences for rigorous competition assays. |
| High-Sensitivity Streptavidin-HRP or Fluorescence Detection Kits | Enable detection of faint bands or quantification in solution (FP). |
| Native Gels (Pre-cast or Hand-cast) | Must provide optimal pore size for resolving large supershifted complexes. |
| Capillary Electrophoresis Systems with UV/Vis or LIF Detection | For high-throughput, automated, quantitative analysis of binding reactions. |
| 96- or 384-Well Microplates (Black, Low Fluorescence) | Essential for FP-EMSA and other plate-based homogeneous assay formats. |
| Mobility Shift Buffer Systems (with/without additives) | Optimized buffers containing carrier proteins (BSA), non-specific competitors (poly dI-dC), and stabilizing agents. |
Diagram: High-Throughput EMSA Evolution
In conclusion, while "EMSA" and "gel retardation assay" are often used interchangeably, the advanced applications of supershifts, competition assays, and high-throughput variations represent the maturation of this technique. These methods provide detailed mechanistic insights and scalable platforms that are indispensable in modern molecular biology and drug development research focused on protein-nucleic acid interactions.
Solving Common EMSA Problems: Expert Tips for Optimization and Clear Results
This technical guide addresses the persistent challenge of weak or absent electrophoretic mobility shifts in nucleic acid-protein interaction studies. Framed within the critical thesis differentiating the Electrophoretic Mobility Shift Assay (EMSA) from the broader, sometimes synonymous, gel retardation assay, this document focuses on core experimental variables. While the terms are often used interchangeably, a key distinction lies in the EMSA's specific use of labeled probes for quantitative analysis of defined complexes, whereas "gel retardation" can describe a wider set of techniques observing mobility changes. Precise troubleshooting is therefore paramount for reliable, publication-quality EMSA data.
Table 1: Impact of Protein Purity on Shift Intensity
| Protein Purity Method | Typical Shift Intensity (Relative) | Key Contaminant Risks |
|---|---|---|
| Crude Lysate | 1.0 (Baseline) | Nucleases, competitor proteins |
| Affinity Purification (Tag) | 4.0 - 8.0 | Protease contamination |
| Ion-Exchange Chromatography | 3.0 - 6.0 | High salt concentration |
| Size-Exclusion Chromatography | 2.0 - 5.0 | Dilution, buffer exchange |
Table 2: Probe Integrity Metrics and Outcomes
| Probe Quality Metric | Acceptable Threshold | Effect on Shift |
|---|---|---|
| Specific Activity (cpm/µl) | > 10,000 | Directly correlates with signal |
| % Incorporation (Labeling) | > 70% | Low = weak/no shift |
| Degradation (Visualized on gel) | Single, sharp band | Smear = non-specific binding/background |
| Molar Excess (Protein:Probe) | 1:1 to 10:1 (optimal) | Too high = probe depletion artifacts |
Detailed Experimental Protocols
Protocol 1: Assessing Nuclear Extract Protein Quality for EMSA
Objective: To isolate and qualify nuclear proteins competent for sequence-specific DNA binding.
- Harvest cells and pellet. Resuspend in 5x pellet volume of hypotonic buffer (10 mM HEPES pH 7.9, 1.5 mM MgCl₂, 10 mM KCl, 0.5 mM DTT, protease inhibitors) on ice for 10 min.
- Centrifuge at 4°C, 850 x g for 10 min. Discard supernatant.
- Resuspend cell pellet in 2x pellet volume of hypotonic buffer. Lyse cells with 10-15 strokes in a Dounce homogenizer (tight pestle).
- Centrifuge nuclei at 4°C, 850 x g for 10 min.
- Resuspend nuclear pellet in high-salt extraction buffer (20 mM HEPES pH 7.9, 25% glycerol, 1.5 mM MgCl₂, 0.8 M KCl, 0.5 mM DTT, protease inhibitors). Rock gently at 4°C for 30-60 min.
- Centrifuge at 4°C, >20,000 x g for 30 min. Aliquot supernatant (nuclear extract), flash-freeze in LN₂, store at -80°C.
- Quality Control: Determine protein concentration (Bradford assay). Run 5-10 µg on SDS-PAGE and Coomassie stain to check for histone enrichment (bands at ~15-20 kDa). Test functionality via EMSA with a known, high-affinity consensus probe.
Protocol 2: Purifying and Quantifying Labeled Oligonucleotide Probe
Objective: Generate a high-specific-activity, double-stranded probe free of unincorporated nucleotides.
- Anneal complementary single-stranded oligonucleotides (10 µM each in 10 mM Tris, 50 mM NaCl, 1 mM EDTA) by heating to 95°C for 5 min and cooling slowly to room temperature.
- Label 50-100 ng of duplex probe using T4 Polynucleotide Kinase and [γ-³²P]ATP (or a non-radioactive end-labeling system) in 1X PNK buffer for 30 min at 37°C.
- Remove unincorporated nucleotides using a spin column (e.g., Sephadex G-25) per manufacturer instructions. Alternative: Purify via native polyacrylamide gel electrophoresis, excise the band, and elute overnight.
- Quantification: Measure radioactivity of 1 µl of purified probe by scintillation counting. Calculate specific activity (cpm/µl). Analyze 10,000 cpm on a 6% native polyacrylamide gel, expose to a phosphorimager screen; a single, tight band should be visible.
Protocol 3: Optimizing Binding Reaction Conditions
Objective: Systematically vary critical parameters to maximize specific complex formation.
- Master Mix Setup: Prepare a master mix containing buffer (10 mM Tris, 50-100 mM KCl/NaCl, 1 mM DTT, 1 mM EDTA, 5% glycerol, pH 7.5), poly(dI-dC) (0.05-0.1 µg/µL), and purified protein (5-20 fmol).
- Competition Setup: For specificity tests, include a 50-100x molar excess of unlabeled specific or non-specific competitor DNA in the master mix. Pre-incubate with protein for 10 min on ice before adding labeled probe.
- Probe Addition: Add 1-5 fmol ( ~10,000 cpm) of labeled probe to each reaction. Final reaction volume: 20 µL.
- Incubation: Incubate at 20-25°C for 20-30 minutes. Key Variables to Titrate: Salt concentration (50-150 mM KCl), divalent cation presence/absence (0-5 mM MgCl₂), carrier DNA type/concentration, glycerol concentration (0-10%), non-ionic detergents (0-0.1% NP-40).
- Electrophoresis: Load entire reaction on a pre-run 6% native polyacrylamide gel (0.5X TBE, 4°C). Run at 100-150 V until dye front migrates appropriate distance.
Diagrams
Title: EMSA Troubleshooting Decision Pathway
Title: Standard EMSA Experimental Workflow
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for EMSA
| Reagent/Material | Function & Purpose | Critical Notes |
|---|---|---|
| Poly(dI-dC) | Non-specific carrier DNA. Competes for non-sequence-specific protein binding to the probe, reducing background. | Concentration must be titrated (0.05-0.5 µg/µL). Too much can disrupt specific binding. |
| [γ-³²P]ATP or Chemiluminescent Labeling Kit | Provides radioactive or non-radioactive tag for probe detection. | ³²P offers high sensitivity; chemiluminescent kits are safer and stable longer. |
| T4 Polynucleotide Kinase (PNK) | Catalyzes transfer of phosphate group to 5' hydroxyl terminus of DNA/RNA. Essential for end-labeling. | Use fresh DTT in reaction buffer for optimal activity. |
| Non-ionic Detergent (e.g., NP-40, Tween-20) | Added to binding buffer (0.01-0.1%). Reduces protein-protein and protein-tube non-specific adhesion. | Avoid SDS or high concentrations that denature proteins. |
| Protease & Phosphatase Inhibitor Cocktails | Preserves protein integrity and post-translational modification state during extraction and binding. | Essential for studying modified transcription factors. Must be added fresh. |
| High-Purity BSA or Gelatin | Alternative carrier protein. Stabilizes dilute protein solutions and can block non-specific binding. | Use nuclease-free, acetylated BSA. Not always beneficial; requires testing. |
| Cold Competitor Oligonucleotides | Unlabeled DNA fragments. Validate binding specificity (specific competitor) and demonstrate sequence specificity (non-specific/mutant competitor). | Critical control. Must be identical to probe sequence (specific) or contain scrambled/mutant sites. |
This whitepaper addresses a critical experimental optimization challenge within the broader thesis research comparing Electrophoretic Mobility Shift Assays (EMSA) and classic gel retardation assays. While often used interchangeably, a key distinction in our thesis framework is that "gel retardation" refers to the foundational observation of protein-nucleic acid complex migration shift, while "EMSA" denotes the modern, optimized protocol incorporating specific strategies to enhance sensitivity and specificity. A principal differentiator is the systematic use of non-specific competitors—like poly(deoxyinosinic-deoxycytidylic) acid (poly dI:dC) and non-specific DNA (NS DNA)—to resolve high background and non-specific binding, which is more rigorously characterized in contemporary EMSA. This guide details their role as a cornerstone of reliable assay execution.
Non-specific binding in EMSA/gel retardation assays arises from electrostatic interactions between positively charged domains in the protein preparation (e.g., histones, contaminating proteins) and the negatively charged phosphate backbone of the probe DNA or RNA. This results in:
- Diffuse or smeared bands on the autoradiogram.
- Multiple shifted bands not competed by cold specific oligonucleotide.
- High background radioactivity in the gel lane, obscuring specific retarded complexes.
- Poor signal-to-noise ratio, compromising quantification.
The Solution: Mechanism of Action of Competitors
Competitors are inert, non-specific nucleic acids added in excess to the binding reaction. They act as "decoys," absorbing the non-specific binding capacity of the protein extract. This allows the labeled, sequence-specific probe to interact primarily with its cognate DNA-binding protein.
- Poly dI:dC: A synthetic, double-stranded polynucleotide with irregular purine-pyrimidine sequences. Its lack of a defined sequence makes it an effective scavenger for a broad range of non-specific DNA-binding proteins. It is particularly effective against proteins with affinity for generic DNA structures.
- Non-Specific DNA: Often sheared or sonicated genomic DNA (e.g., from salmon sperm or E. coli) or unrelated oligonucleotides (e.g., a mutated version of the target site). It competes for proteins binding to DNA in a sequence-independent manner.
Quantitative Data and Optimization
The optimal type and amount of competitor are empirical and must be titrated for each new protein extract and probe. The following table summarizes key optimization data from recent literature and protocols.
Table 1: Competitor Optimization Guide for EMSA
| Competitor Type | Typical Working Concentration Range (per 20µL reaction) | Effective Against | Potential Pitfalls if Overused |
|---|---|---|---|
| Poly dI:dC | 0.05 µg – 2 µg | General non-specific DNA-binding proteins, histone contaminants. | Can compete for specific protein if used in extreme excess; may inhibit formation of specific complex. |
| Sonicated Salmon Sperm DNA | 0.1 µg – 4 µg | Broad-spectrum non-specific binders; useful for crude nuclear extracts. | Can be less effective per microgram than poly dI:dC for some targets; may contain bioactive contaminants. |
| Non-Specific Oligonucleotide | 50-fold – 500-fold molar excess over probe | Proteins with weak sequence preference similar to target. | Requires careful design to ensure no specific binding sites are present. |
| Carrier tRNA / Yeast RNA | 0.1 µg – 1 µg | RNA-binding proteins (in RNA-EMSA); reduces non-specific protein-RNA interactions. | Irrelevant for standard DNA-EMSA. |
Table 2: Example Titration Experiment Results for a Nuclear Extract & NF-κB Probe
| Poly dI:dC Added (µg) | Specific Complex Intensity | Background/Smear | Recommended? |
|---|---|---|---|
| 0 | Very Low | Very High | No |
| 0.1 | Low | High | No |
| 0.25 | High | Low | Yes |
| 0.5 | Medium | Very Low | Possibly |
| 1.0 | Low | Absent | No (Specific binding competed) |
Detailed Experimental Protocols
Protocol 1: Standard EMSA Binding Reaction with Competitor Optimization
Objective: To establish the optimal amount of poly dI:dC for a new protein extract. Reagents: Labeled specific probe, unlabeled specific competitor (cold probe), unlabeled non-specific competitor (mutant probe), poly dI:dC stock (1 µg/µL), nuclear extract, EMSA binding buffer (10 mM HEPES pH 7.9, 50 mM KCl, 1 mM DTT, 2.5 mM MgCl₂, 0.1 mM EDTA, 10% glycerol), polyacrylamide gel. Procedure:
- Prepare a master mix containing buffer, labeled probe (20,000 cpm), and protein extract (5-10 µg). Keep on ice.
- Aliquot the master mix into 5 tubes.
- Add poly dI:dC to final amounts of 0, 0.1, 0.25, 0.5, and 1.0 µg per tube. Adjust volume with buffer.
- Include control reactions: one with a 100-fold molar excess of unlabeled specific competitor, and one with a 100-fold excess of non-specific competitor.
- Incubate at room temperature for 20 minutes.
- Load directly onto a pre-run, non-denaturing 4-6% polyacrylamide gel in 0.5x TBE.
- Run gel at 100V at 4°C until dye front migrates appropriately.
- Dry gel and expose to phosphorimager screen or X-ray film.
Protocol 2: Supershift EMSA with Competitors
Objective: To identify a protein in a specific complex while maintaining low background. Reagents: As in Protocol 1, plus antibody against the suspected DNA-binding protein. Procedure:
- Set up the optimal binding reaction as determined in Protocol 1 (including the ideal poly dI:dC amount).
- After the initial 20-minute incubation, add 1-2 µg of specific antibody or an equivalent amount of control IgG.
- Incubate for an additional 30-60 minutes at 4°C to allow antibody-protein complex formation.
- Load and run gel as in Protocol 1. Note: The antibody-protein-DNA complex ("supershift") will migrate even slower than the original protein-DNA complex.
Visualizations
Mechanism of Competitor Action in EMSA
EMSA Competitor Optimization Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Resolving Non-Specific Binding in EMSA
| Reagent | Function & Rationale | Example Product/Catalog Note |
|---|---|---|
| Poly dI:dC | Synthetic, sequence-random competitor. Gold standard for absorbing non-specific DNA-binding proteins in crude extracts. | MilliporeSigma (P4929), Thermo Fisher Scientific (20148E). Store aliquots at -20°C. |
| Sonicated Salmon Sperm DNA | Natural DNA sheared to ~500 bp. Alternative broad-spectrum competitor, often used in combination with poly dI:dC. | Thermo Fisher Scientific (15632011). Requires heat-denaturation and quick chilling before use. |
| Non-Specific/ Mutant Oligonucleotide | Unlabeled duplex oligonucleotide with a mutated binding site. Controls for sequence specificity more precisely than random polymers. | Designed in-house and ordered from any oligo synthesis service. Use at 50-200x molar excess. |
| Non-Radioactive Probe Labeling Kits | (e.g., Biotin, DIG, Fluorescence). Reduces safety concerns and allows rapid detection. Competitor requirements are identical to radioactive EMSA. | Thermo Fisher Scientific LightShift Chemiluminescent EMSA Kit (20148). |
| Protease & Phosphatase Inhibitor Cocktails | Added to protein extraction buffers. Prevents degradation and preserves modification states of the target protein, ensuring a cleaner specific signal. | Roche cOmplete EDTA-free (5056489001), PhosSTOP (4906845001). |
| High-Purity Bovine Serum Albumin (BSA) or Casein | Added to binding buffer (to 0.1-0.5 mg/mL). Blocks non-specific protein adsorption to tubes and stabilizes the binding reaction. | New England Biolabs (B9000S - BSA). |
Within the context of a broader thesis investigating the nuanced differences between Electrophoretic Mobility Shift Assays (EMSA) and gel retardation assays, optimizing native polyacrylamide gel electrophoresis (PAGE) conditions is paramount. While the terms are often used interchangeably, a precise thesis distinguishes EMSA as a specific application for detecting protein-nucleic acid interactions, whereas gel retardation is a broader phenomenon. This technical guide details the core parameters governing resolution, reproducibility, and data validity in these critical techniques.
Resolving Power: Gel Composition and Matrix Parameters
Resolving power defines the ability to distinguish between protein-nucleic acid complexes and free probes, or between complexes of different stoichiometry. The primary lever is the polyacrylamide gel matrix.
Key Parameters:
- Acrylamide:Bis-acrylamide Ratio: Typically 29:1 or 37.5:1 for native gels. A higher crosslinker ratio (e.g., 29:1) creates a tighter mesh, better for resolving smaller complexes.
- Total Percentage (%T): Ranges from 4-10% for native EMSA. Lower percentages (4-6%) resolve large complexes; higher percentages (8-10%) resolve smaller complexes and free probes.
- pH of Polymerization: Affects pore formation. Consistent, neutral pH (7.0-7.5) is standard.
Table 1: Optimized Gel Composition for Complex Resolution
| Target Complex Size (kDa) | Recommended %T | Acrylamide:Bis Ratio | Expected Resolution Outcome |
|---|---|---|---|
| Large (>200 kDa) | 4% | 29:1 | Separation of large complex from mid-size; free probe may run off. |
| Medium (50-200 kDa) | 6% | 37.5:1 or 29:1 | Optimal balance for complex vs. free probe separation. |
| Small (<50 kDa) & Free Probe | 8-10% | 37.5:1 | Sharp bands for free probe and small shifts; large complexes may not enter. |
Protocol: Casting a Standard 6% Native Polyacrylamide Gel
- Clean glass plates and spacers (1.0-1.5 mm) thoroughly.
- Prepare 10 mL of gel solution:
- 1.5 mL 40% Acrylamide/Bis solution (29:1)
- 1.0 mL 10X TBE (or TAE) buffer
- 7.4 mL deionized water
- 50 µL 10% Ammonium Persulfate (APS)
- 10 µL Tetramethylethylenediamine (TEMED)
- Mix quickly, pour between plates, insert comb, and allow to polymerize for 30-45 min at room temperature.
Electrophoresis Parameters: Field Strength, Temperature, and Time
Electrophoresis conditions must preserve non-covalent complexes while achieving separation.
Key Parameters:
- Buffer Ionic Strength: Low ionic strength (0.25-0.5X TBE) reduces heat generation but can destabilize some complexes. 0.5X TBE is a common compromise.
- Voltage/Field Strength: High voltage causes heating, leading to complex dissociation ("band smearing") and buffer pH shifts. A constant 80-150 V (≈10 V/cm) is recommended.
- Temperature: Run in a cold room (4°C) or with active cooling to minimize heat-induced dissociation.
- Run Time: Determined empirically; run until the free probe (tracked by dye front) is 2/3 to 3/4 down the gel.
Table 2: Electrophoresis Conditions for Complex Stability
| Parameter | Recommended Setting | Technical Rationale | Risk of Deviation |
|---|---|---|---|
| Buffer | 0.5X TBE | Adequate conductivity with moderate heat. | High ionic strength: overheating. Low ionic strength: complex destabilization. |
| Voltage | 100 V constant | Maintains ~10 V/cm for standard mini-gel. | >150 V: significant joule heating, band smearing. |
| Temperature | 4°C (active cooling) | Minimizes complex dissociation kinetics. | Room temp: increased dissociation, poor band definition. |
| Run Duration | ~1.5-2 hours (until dye front is 3/4 down) | Sufficient separation window. | Too short: poor resolution. Too long: band diffusion. |
Protocol: Electrophoresis Run Setup
- Place polymerized gel into electrophoresis tank.
- Fill upper and lower chambers with pre-chilled 0.5X TBE running buffer.
- Carefully load samples (mixed with non-denaturing loading dye) into wells.
- Run at constant 100 V in a cold room (4°C) or with a circulating cooling unit.
- Stop when bromophenol blue dye front has migrated to the bottom 1/4 of the gel.
- Proceed to transfer (for blotting) or staining.
Buffer Stability: Consistency Across Runs
Buffer degradation, ion depletion (especially for Tris-based buffers), and pH drift are major sources of inter-experimental variability.
Key Parameters:
- Buffer Re-circulation: For runs >2 hours, buffer recirculation between anode and cathode chambers prevents ion gradient formation.
- Buffer Refresh Policy: Replace tank buffer after 2-3 runs to maintain consistent pH and ionic strength.
- pH Monitoring: Check running buffer pH before critical experiments; it should be 8.3 for TBE.
Table 3: Buffer Management Protocol
| Buffer Component | Function | Stability Concern | Mitigation Strategy |
|---|---|---|---|
| Tris-HCl | pH buffer | Anode: pH decreases (acidification). Cathode: pH increases. | Use buffer with adequate capacity (0.5X), implement recirculation, refresh frequently. |
| Boric Acid / Acetate | Conductivity & buffering | Borate can form complexes with cis-diols. | For RNA probes, consider Tris-Glycine or HEPES-based buffers. |
| EDTA | Divalent cation chelator | Oxidation over time. | Prepare fresh from stock or use frozen aliquots. |
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for EMSA/Gel Retardation Assays
| Item | Function & Specification | Critical Note |
|---|---|---|
| High-Purity Acrylamide/Bis | Gel matrix formation. Use electrophoresis-grade, >99.9% purity. | Impurities inhibit polymerization and increase background. |
| TEMED & APS (Persulfate) | Redox initiators for gel polymerization. Use fresh APS solution (<1 week old at 4°C). | Degraded APS leads to uneven or failed polymerization. |
| Non-Denaturing Loading Dye | Sample visualization during loading without disrupting complexes. Contains glycerol, bromophenol blue, xylene cyanol. | Do not use SDS or β-mercaptoethanol. |
| High-Binding-Affinity Membranes (e.g., Nylon+) | For capillary transfer of nucleic acids in blot-based detection. Positively charged for nucleic acid retention. | Nitrocellulose has lower binding capacity for small oligonucleotides. |
| Chemiluminescent Substrate (e.g., ECL-based) | For high-sensitivity detection of horseradish peroxidase (HRP)-labeled probes. | More sensitive than colorimetric detection for low-abundance complexes. |
| Cold Room or Circulating Cooler | Maintains 4°C during electrophoresis. | Essential for labile protein-DNA/RNA interactions. |
Visualizing the Optimization Workflow and Context
Title: Core Workflow for Gel Optimization in EMSA Research
Title: Molecular Basis of Gel Retardation in EMSA
Addressing Probe Degradation and Handling of Radioactive Materials Safely
The Electrophoretic Mobility Shift Assay (EMSA) and the synonymous gel retardation assay are cornerstone techniques for studying nucleic acid-protein interactions. While the terms are often used interchangeably, a modern thesis might frame "gel retardation" as the broader historical principle and "EMSA" as its standardized, contemporary implementation. This technical guide focuses on a critical, shared vulnerability: the integrity of the nucleic acid probe. Probe degradation leads to high background noise, reduced specific signal, and unreliable quantification, directly confounding research on transcription factors, RNA-binding proteins, and therapeutic targeting. Furthermore, the use of radioactive isotopes (e.g., ³²P) for supreme sensitivity necessitates stringent safety protocols. This whitepaper provides an in-depth guide to mitigating probe degradation and ensuring safe radiological handling within these assays.
Mechanisms and Impact of Probe Degradation
Probe degradation occurs via enzymatic (nucleases) and chemical (hydrolysis, oxidation) pathways. Degraded probes produce shorter fragments that migrate unpredictably or smear, obscuring the discrete shifted band.
Table 1: Common Sources of Probe Degradation and Preventive Measures
| Source of Degradation | Mechanism | Impact on EMSA | Preventive Solution |
|---|---|---|---|
| Ribonucleases (RNases) | Endoribonucleolytic cleavage of RNA probes. | Complete loss of signal, smearing. | Use RNase inhibitors (e.g., RNasin), DEPC-treated water, dedicated RNA workspace. |
| Deoxyribonucleases (DNases) | Endonucleolytic cleavage of DNA probes. | Reduced specific signal, increased background. | Use DNase inhibitors (e.g., EDTA), high-purity reagents. |
| Chemical Hydrolysis | Acid/alkaline cleavage of phosphodiester backbone. | Probe fragmentation. | Maintain neutral pH in buffers, use Tris-EDTA (TE) for storage. |
| Oxidative Damage | Radical-mediated strand breakage. | Reduced binding affinity, fragmentation. | Include antioxidants (e.g., DTT, β-mercaptoethanol) in binding reactions. |
| Improper Storage | Repeated freeze-thaw cycles, elevated temperature. | Cumulative damage from all above. | Store probes in single-use aliquots at -80°C, avoid repeated thawing. |
Detailed Experimental Protocols for Probe Integrity
Protocol: Synthesis and Purification of a Radiolabeled Probe (³²P)
- Materials: DNA oligonucleotide, [γ-³²P]ATP, T4 Polynucleotide Kinase (PNK), NucAway Spin Columns, TE buffer.
- Method:
- In a shielded microcentrifuge tube, mix: 1 µL oligonucleotide (100 ng), 2 µL 10X PNK buffer, 1 µL T4 PNK (10 U), 15 µL nuclease-free water, and 5 µL [γ-³²P]ATP (50 µCi).
- Incubate at 37°C for 30 minutes.
- Terminate reaction by heating at 65°C for 5 minutes.
- Purification: Pre-hydrate a NucAway column. Load the reaction mix onto the column and centrifuge at 12,000g for 2 minutes. The flow-through contains the purified labeled probe. Determine specific activity by scintillation counting.
- Dilute probe in TE buffer (pH 8.0) with 1 mM DTT. Store at -80°C in a lead-lined box.
Protocol: EMSA with Focus on Probe Handling
- Binding Reaction:
- Thaw reagents on ice. Prepare a master mix for n+1 reactions containing: 2 µL 10X Binding Buffer (100 mM Tris, 500 mM KCl, 10 mM DTT), 1 µL Poly(dI:dC) (1 µg/µL), 1 µL labeled probe (~20,000 cpm), and nuclease-free water to 18 µL.
- Aliquot 18 µL of master mix into reaction tubes. Add 2 µL of nuclear extract or purified protein (include a probe-only control).
- Incubate at room temperature for 20-30 minutes.
- Gel Electrophoresis:
- Pre-run a 6% non-denaturing polyacrylamide gel in 0.5X TBE at 100V for 30-60 minutes in a cold room.
- Load samples with 5 µL of non-denaturing loading dye. Run at 80-100V until the dye front is near the bottom (1-2 hours).
- Detection: For radioactive probes, transfer gel to filter paper, dry under vacuum, and expose to a phosphorimager screen overnight.
Safety Protocol for Radioactive Material (³²P) Handling
- Designated Area: Use a dedicated, clearly marked workspace with absorbent bench liners and shielding (≥1 cm thick acrylic for β-emitters like ³²P).
- Personal Protective Equipment (PPE): Lab coat, disposable gloves, safety glasses, and a dosimeter ring.
- Containment: Use dedicated, labeled microcentrifuges and pipettes. All waste must go into clearly labeled radioactive waste containers (solid/liquid).
- Monitoring: Survey work area, equipment, and skin after each procedure with a Geiger-Müller counter. Decontaminate immediately if contamination is found.
- Hygiene: Strictly enforce a no-eating, no-drinking policy. Perform a final full-body survey before leaving the lab.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Robust EMSA
| Item | Function & Importance |
|---|---|
| RNase Inhibitor (e.g., RNasin) | Inactivates ribonucleases by non-competitive binding, critical for RNA probe and RBP studies. |
| High-Specific-Activity [γ-³²P]ATP | Provides the sensitivity needed to detect low-abundance protein complexes. Non-radioactive alternatives (chemiluminescence) are less sensitive. |
| T4 Polynucleotide Kinase (PNK) | Catalyzes the transfer of the terminal phosphate from ATP to the 5'-OH of nucleic acids for labeling. |
| Non-Specific Competitor DNA (Poly(dI:dC)) | Binds to non-specific proteins, reducing background and clarifying specific shifted bands. |
| Dithiothreitol (DTT) | A reducing agent that maintains protein sulfhydryl groups and acts as an antioxidant, protecting probe integrity. |
| NucAway or G-25 Spin Columns | Remove unincorporated nucleotides, ensuring clean signal and accurate quantification. |
| Acrylamide/Bis-Acrylamide (29:1) | Forms the matrix of the non-denaturing gel, essential for separating protein-nucleic acid complexes from free probe. |
Visualizations
Title: EMSA Workflow and Probe Degradation Threats
Title: Safe Radioactive Probe Handling Workflow
Within the broader thesis on distinguishing Electrophoretic Mobility Shift Assays (EMSA) from gel retardation assays, a critical intersection emerges: the need for rigorous, standardized methodologies to ensure data reproducibility. While often used interchangeably, purists argue "EMSA" implies a specific, quantitative detection method, whereas "gel retardation" is a broader descriptive term. This technical guide addresses the shared experimental core and the controls essential for generating reliable, interpretable data in both frameworks, crucial for researchers and drug development professionals investigating transcription factors, nucleic acid-protein interactions, and gene regulation therapeutics.
Foundational Concepts: EMSA vs. Gel Retardation
At its core, both assays detect complexes between proteins and nucleic acids by observing a reduction in electrophoretic mobility (a "shift") in a non-denaturing gel. The historical and methodological nuances are summarized below.
Table 1: Comparative Analysis of EMSA and Gel Retardation Assay Contexts
| Aspect | Classical Gel Retardation Assay | Modern Quantitative EMSA |
|---|---|---|
| Primary Definition | Descriptive term for the observed phenomenon of reduced mobility. | A specific technique employing labeled probes for quantitative analysis. |
| Detection Method | Often ethidium bromide staining (for DNA/RNA), less sensitive. | Relies on radioactive (³²P) or fluorescent/chemiluminescent labeled probes. |
| Data Output | Qualitative or semi-quantitative; confirms binding presence. | Quantitative; can determine dissociation constants (Kd), stoichiometry. |
| Reproducibility Challenge | High variability due to stain intensity, loading inconsistencies. | Higher inherent reproducibility but susceptible to protocol deviations. |
Standardized Protocol for a Quantitative EMSA
The following protocol is optimized for reproducibility and quantitative analysis, suitable for investigating specific DNA-protein interactions.
I. Reagent Preparation
- Binding Buffer (10X Stock): 100 mM HEPES (pH 7.9), 400 mM KCl, 10 mM DTT, 10 mM EDTA, 50% Glycerol. Store at -20°C. Critical: Aliquot to avoid freeze-thaw cycles of DTT.
- Poly(dI-dC): A nonspecific competitor DNA. Concentration must be titrated for each new protein extract (typical range: 0.05-0.5 µg/µL per reaction).
- Labeled Probe: 5'-end labeled double-stranded oligonucleotide (20-40 bp) containing the protein binding site. Purify via native PAGE or column purification. Specific activity > 107 cpm/µg is ideal for detection.
- Non-Denaturing Polyacrylamide Gel (6%): 29:1 acrylamide:bis-acrylamide in 0.5X TBE. Pre-run for 60 min at 100V before loading samples.
II. Binding Reaction Assembly (20 µL total volume)
Perform in low-protein-binding tubes. Include controls as defined in Section III.
- Master Mix (per reaction): 2 µL 10X Binding Buffer, 1 µL Poly(dI-dC) (at optimized concentration), 1 µL 1 mg/mL BSA, x µL Nuclease-free H2O.
- Add 2 µL of purified nuclear extract or recombinant protein (containing 2-10 µg total protein). Include a "No Protein" control lane.
- Add 1 µL of labeled probe (~20 fmol, 20,000-50,000 cpm).
- Incubate at 25°C for 30 minutes.
- Optional Supershift: Add 1-2 µL of specific antibody after 20 min, incubate further 10-20 min.
III. Electrophoresis and Detection
- Add 5 µL of non-denaturing loading dye (0.25% bromophenol blue, 30% glycerol) to each reaction. Do not heat.
- Load entire sample onto pre-run 6% non-denaturing gel in 0.5X TBE buffer.
- Run at 100V constant voltage at 4°C until dye front is ~2/3 down the gel (time varies by apparatus).
- Transfer gel to chromatography paper, dry under vacuum, and expose to phosphorimager screen or X-ray film. For fluorescent probes, image using appropriate scanner.
The Critical Role of Controls in Experimental Design
Table 2: Essential Positive and Negative Controls for EMSA/Gel Retardation
| Control Type | Purpose | Composition | Expected Result | Interpretation of Failure |
|---|---|---|---|---|
| Negative Control 1: No Protein | Baseline for free probe migration. | Reaction without protein/extract. | Single band at free probe position. | Probe degradation or gel artifacts if smear/no band. |
| Negative Control 2: Nonspecific Competitor | Confirms sequence-specific binding. | Reaction + 100-fold molar excess of unlabeled nonspecific DNA (e.g., mutated site). | Shifted band persists. | Loss of shift indicates non-specific binding. |
| Positive Control 1: Specific Competitor (Cold Probe) | Confirms binding specificity and identity. | Reaction + 100-fold molar excess of unlabeled specific probe (identical sequence). | Significant reduction/abolition of shifted band. | Shift persists: complex is not specific to intended sequence. |
| Positive Control 2: Known Active Extract | Validates assay system functionality. | Reaction using a protein/extract with known, validated binding activity. | Clear shifted band. | No shift: assay reagents (buffer, gel, probe label) are faulty. |
| Antibody Supershift | Identifies protein in complex. | Reaction + antibody against suspected DNA-binding protein. | Further retardation ("supershift") or ablation of complex. | No effect: protein not present or epitope masked. |
| Mutant Probe Control | Defines exact sequence requirement. | Reaction with labeled probe containing a point mutation in the binding site. | No shifted band formed. | Shift occurs: protein binds non-specifically or mutation is not critical. |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Reproducible EMSA
| Item | Function & Importance | Reproducibility Tip |
|---|---|---|
| Chemiluminescent Nucleic Acid Labeling Kit | Non-radioactive, safe labeling of DNA probes. High sensitivity. | Use fresh labeling reagents; validate probe specific activity for each batch. |
| High-Purity Acrylamide/Bis-acrylamide (29:1) | Matrix for non-denaturing gel electrophoresis. | Prepare fresh from powder or use certified pre-mixed solutions; degas before polymerization. |
| Recombinant Protein or Characterized Nuclear Extract | Source of DNA-binding protein. | Use aliquots from a single, well-characterized preparation; document protein concentration and buffer. |
| Carrier DNA (Poly(dI-dC)) | Suppresses non-specific protein-nucleic acid interactions. | Titrate for each new protein preparation; excess can disrupt specific binding. |
| Dithiothreitol (DTT) | Reducing agent maintaining protein sulfhydryl groups. | Prepare fresh 1M stock in water; aliquot and store at -20°C; add to buffer just before use. |
| Phosphorimager System | Quantitative digital imaging of radioactive/chemiluminescent signals. | Regular calibration and linear range validation are essential for quantitation. |
| Gel Drying Apparatus | Prepares gel for autoradiography. | Ensure even heating to prevent gel cracking, which ruins the experiment. |
| Low-Binding Microcentrifuge Tubes | Minimizes protein loss to tube walls. | Use consistently; do not substitute with standard tubes. |
Visualization of Workflows and Logic
Title: EMSA Experimental Workflow with Quality Checkpoints
Title: Interdependence of EMSA Controls for Interpretation
Supershift assays, a sophisticated variant of the Electrophoretic Mobility Shift Assay (EMSA), are pivotal in identifying specific protein components within a nucleic acid-protein complex. Within the broader thesis distinguishing EMSA (a technique focused on detecting any binding event) from the more general "gel retardation assay" (a term sometimes used synonymously but often implying a simpler confirmation of binding), the supershift assay stands out as a definitive tool for complex deconvolution. Its success, however, is critically dependent on two interlinked factors: the quality of the antibody and the accessibility of its target epitope.
The Critical Role of Antibody Quality
A high-affinity, specific antibody is non-negotiable. Key quantitative parameters that define antibody quality include:
Table 1: Quantitative Metrics for Supershift Antibody Evaluation
| Metric | Optimal Range | Impact on Supershift Assay |
|---|---|---|
| Affinity Constant (K_D) | ≤ 1 nM | Drives efficient complex formation during the short incubation period. |
| Titer (ELISA) | ≥ 1:100,000 | Indicates high abundance of specific antibody, reducing non-specific background. |
| Cross-Reactivity | < 5% vs. related isoforms | Ensures the supershift is specific to the target protein, not a family member. |
| Endotoxin Level | < 1 EU/mg | Prevents non-specific protein aggregation or complex disruption. |
Epitope Accessibility: A Structural Challenge
Even an excellent antibody may fail if its epitope is obscured. Inaccessibility can result from:
- Protein-Protein Interactions: The epitope is buried within the DNA-protein or protein-protein interface.
- Conformational Change: DNA binding induces a structural shift, hiding the epitope.
- Post-Translational Modifications (PTMs): Phosphorylation or other PTMs can alter epitope structure.
Experimental Protocols for Systematic Troubleshooting
Protocol 1: Pre-Validation of Antibody for Supershift
- Western Blot: Verify specificity against nuclear extract. A single band at the correct molecular weight is ideal.
- Immunoprecipitation (IP)-EMSA: Incubate antibody with nuclear extract, pull down the complex, and then use the eluate in a standard EMSA. A positive result confirms the antibody binds the native protein.
- Peptide Blocking Control: Pre-incubate antibody with a 10-fold molar excess of the immunizing peptide for 1 hour at 4°C before adding to the EMSA binding reaction. The supershift should be abolished.
Protocol 2: Assessing Epitope Accessibility via Altered Binding Order
- Standard Method: Add antibody after the DNA-protein complex is formed (20-min incubation post-binding reaction).
- Alternative Method 1: Pre-incubate antibody with the protein extract for 20 min at 4°C before adding the labeled probe. This can capture the protein before DNA-induced occlusion.
- Alternative Method 2: Add antibody simultaneously with the probe to the protein extract. This tripartite co-incubation may allow complex capture in a different conformation. Compare the efficiency of supershift formation across these three conditions.
Protocol 3: Modifying Buffer Conditions to Unmask Epitopes
- Salt Variation: Perform binding reactions with KCl or NaCl concentrations ranging from 50 mM to 300 mM. Higher ionic strength can weaken protein-DNA interactions, potentially exposing epitopes.
- Mild Detergent: Include 0.1% NP-40 or Tween-20 in the binding reaction to reduce non-specific aggregation without disrupting specific interactions.
- Carrier Protein: Increase the concentration of nonspecific carrier (e.g., BSA to 0.1 mg/mL) to minimize antibody sticking to tubes.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Robust Supershift Assays
| Reagent | Function & Rationale |
|---|---|
| High-Affinity, Monoclonal Antibody | Superior specificity for a single epitope reduces off-target binding and background. |
| Phospho-Specific or Isoform-Specific Antibodies | For discriminating between modified states or family members within a complex. |
| Control IgG (Isotype-matched) | Critical negative control to identify non-specific supershift artifacts. |
| Immunizing Peptide | For competition controls to definitively prove supershift specificity. |
| Non-ionic Detergent (e.g., NP-40) | To reduce non-specific hydrophobic interactions without denaturing proteins. |
| Protease & Phosphatase Inhibitor Cocktails | Preserves protein integrity and modification states during extract preparation. |
| Chemiluminescent Nucleic Acid Label Detection Kit | Offers higher sensitivity for detecting faint supershifted bands vs. traditional radioisotopes. |
Visualizing Supershift Outcomes and Strategies
Beyond EMSA: Validating and Comparing Results with Complementary Techniques
The Electrophoretic Mobility Shift Assay (EMSA), also historically and interchangeably referred to as the gel retardation assay, is a cornerstone technique in molecular biology for detecting protein-nucleic acid interactions. This whitepaper provides a critical assessment of the sensitivity and specificity of the modern EMSA. It is framed within the thesis that while "EMSA" and "gel retardation assay" are often used synonymously, contemporary EMSA has evolved into a family of related techniques with significant methodological refinements. These advancements, including the use of supershifts, competition assays, and fluorescent/chemiluminescent probes, have directly impacted the core analytical parameters of sensitivity and specificity, distinguishing it from the classical, simpler gel retardation concept.
Core Principles and Quantitative Performance Metrics
EMSA operates on the principle that a protein-nucleic acid complex migrates more slowly through a non-denaturing polyacrylamide or agarose gel than the free nucleic acid probe. The degree of retardation ("shift") is indicative of binding.
The performance of EMSA is quantitatively defined by its sensitivity and specificity, which are in constant trade-off based on experimental design.
Table 1: Key Quantitative Performance Metrics of EMSA
| Parameter | Typical Range | Definition & Impact |
|---|---|---|
| Sensitivity (Detection Limit) | 1-10 fmol of protein | The minimum amount of protein or complex detectable. Depends on probe specific activity, exposure time, and background. |
| Equilibrium Dissociation Constant (Kd) | 10^-9 to 10^-12 M | Can be approximated by EMSA, but is influenced by gel electrophoresis conditions (non-equilibrium). |
| Probe Specific Activity | > 5 x 10^8 cpm/µg (³²P) | Critical for sensitivity. Higher specific activity lowers detection limit. |
| Non-Specific Competitor DNA (e.g., poly(dI-dC)) | 0.1-5 µg/reaction | Used to improve specificity by quenching non-specific protein binding. Optimal amount is protein-specific. |
| Signal-to-Noise Ratio | Variable | Ratio of shifted band intensity to free probe or background. Defines assay robustness. |
Table 2: Strengths and Limitations of EMSA
| Strengths | Limitations |
|---|---|
| Conceptually Simple: Easy to set up and interpret. | Low Throughput: Labor-intensive, not suited for screening. |
| Probe Flexibility: Can use DNA, RNA, or modified nucleotides. | Non-Quantitative for Kd: Electrophoresis disturbs equilibrium; data is semi-quantitative. |
| Functional Specificity: Competition assays provide strong evidence for sequence-specific binding. | Low Sensitivity: Compared to methods like SPR or FP; requires abundant protein. |
| Complex Analysis: Can detect multiple complexes, cooperativity, and stoichiometry. | False Negatives: Binding may be too weak or transient to survive electrophoresis. |
| Antibody "Supershift": Confirms protein identity, enhancing specificity. | False Positives: Non-specific interactions can cause shifts. |
| No Specialized Equipment: Requires standard gel electrophoresis apparatus. | Gel Artifacts: Probe degradation, protein aggregation, or gel irregularities can confound results. |
Detailed Experimental Protocols
Protocol 1: Standard EMSA for a DNA-Binding Protein (e.g., Transcription Factor)
1. Probe Labeling (End-Labeling with [γ-³²P]ATP)
- Materials: Double-stranded oligonucleotide containing consensus sequence, T4 Polynucleotide Kinase (PNK), [γ-³²P]ATP, NucAway spin column.
- Method: In a 20 µL reaction, combine 100 ng DNA, 1X PNK buffer, 20 units PNK, and 50 µCi [γ-³²P]ATP. Incubate at 37°C for 45 min. Heat-inactivate at 65°C for 10 min. Purify labeled probe using spin column to remove unincorporated nucleotides.
2. Binding Reaction
- Materials: Nuclear protein extract or purified protein, labeled probe, poly(dI-dC), binding buffer (10 mM HEPES pH 7.9, 50 mM KCl, 0.5 mM DTT, 0.1 mM EDTA, 5% glycerol).
- Method: Pre-incubate 5-20 µg protein extract with 2 µg poly(dI-dC) in binding buffer (total volume 18 µL) on ice for 10 min. Add 1 µL of labeled probe (~20,000 cpm). Incubate at room temperature for 25 min.
3. Electrophoresis and Detection
- Materials: 6% non-denaturing polyacrylamide gel (29:1 acrylamide:bis), 0.5X TBE running buffer.
- Method: Pre-run gel at 100V for 60 min in a cold room (4°C). Load samples (add 2 µL of 10X loading dye) directly onto running gel. Run at 100V for 60-90 min until dye front migrates 2/3 down. Transfer gel to Whatman paper, dry, and expose to a phosphorimager screen overnight.
Protocol 2: Specificity Control - Cold Competition Assay
- Method: Perform binding reaction as above, but add an increasing molar excess (e.g., 10x, 50x, 100x) of unlabeled "cold" competitor oligonucleotide to the pre-incubation mix before adding the labeled probe. A specific competitor (identical sequence) should abolish the shift. A mutant competitor (scrambled sequence) should not.
Protocol 3: Specificity Confirmation - Antibody Supershift Assay
- Method: After the standard binding reaction, add 1-2 µg of antibody specific to the suspected DNA-binding protein. Incubate further for 30-60 min on ice. The antibody-protein-DNA complex will migrate even slower ("supershift") or may be disrupted, confirming protein identity.
Visualizations
EMSA Core Workflow
Interpreting EMSA Results & Controls
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for EMSA
| Reagent/Material | Function & Rationale |
|---|---|
| High-Activity [γ-³²P]ATP or Fluorescent/Chemiluminescent Probe | Provides the detection signal. Specific activity is paramount for sensitivity. Non-radioactive alternatives reduce hazard. |
| T4 Polynucleotide Kinase (PNK) | Catalyzes the transfer of the terminal phosphate from ATP to the 5'-OH group of DNA/RNA for end-labeling. |
| Purified Protein or High-Quality Nuclear Extract | Source of the DNA/RNA-binding protein. Extract quality (no nucleases/degradases) is critical. |
| Non-Specific Competitor DNA (poly(dI-dC), sheared salmon sperm DNA) | Blocks non-specific binding sites on proteins, dramatically improving assay specificity and band sharpness. |
| Non-Denaturing Gel Electrophoresis System | Separates complex from free probe based on size/charge without disrupting non-covalent interactions. Low ionic strength buffers (e.g., 0.5X TBE) are typical. |
| Specific and Mutant Unlabeled Competitor Oligonucleotides | Validate binding specificity. The mutant control is essential to rule out non-sequence-specific interactions. |
| Antibody for Target Protein | For supershift assays, confirms the identity of the protein in the complex, adding a layer of molecular specificity. |
| Phosphorimager or Fluorescence Gel Scanner | For quantification and visualization of results. Phosphorimagers are far more sensitive than film for radioisotopes. |
Within a broader thesis investigating the differences between Electrophoretic Mobility Shift Assay (EMSA) and gel retardation assays, it is critical to contrast these in vitro techniques with in vivo methodologies. EMSA excels in characterizing protein-nucleic acid interactions in a purified, controlled environment, offering precise biochemical validation. However, to understand the biological context of these interactions—where they occur in the genome, under what conditions, and with what concomitant partners—researchers must employ in vivo methods. Chromatin Immunoprecipitation (ChIP) and its advanced variants represent the gold standard for this purpose, enabling the mapping of protein-DNA interactions within living cells.
Core Technique: Standard Chromatin Immunoprecipitation (ChIP)
Experimental Protocol:
- Crosslinking: Cells/tissues are treated with formaldehyde (typically 1% for 10-15 min at room temperature) to covalently crosslink protein-DNA and protein-protein complexes.
- Cell Lysis & Chromatin Preparation: Cells are lysed, and chromatin is isolated and sheared to 200-1000 bp fragments via sonication or enzymatic digestion (MNase).
- Immunoprecipitation: The sheared chromatin is incubated with a specific antibody against the protein of interest (or an epitope tag). Antibody-chromatin complexes are captured using protein A/G beads.
- Washing & Elution: Beads are stringently washed to remove non-specific background. Crosslinks are reversed (often by heating at 65°C with high salt), and proteins are degraded by Proteinase K.
- DNA Purification & Analysis: Co-precipitated DNA is purified and analyzed. This can be quantitative (qPCR for specific loci) or genome-wide (sequencing, i.e., ChIP-seq).
Key ChIP Variants
ChIP-seq (ChIP followed by sequencing): The dominant genome-wide method. Precipitated DNA is used to generate a sequencing library, allowing for the mapping of all binding sites for the protein of interest across the genome.
CUT&RUN (Cleavage Under Targets and Release Using Nuclease): An in situ alternative. Permeabilized cells are incubated with an antibody, followed by protein A/G fused to micrococcal nuclease (MNase). Activation of MNase cleaves DNA around the target, releasing specific fragments for sequencing. Advantages include lower cell input (~1000 cells), superior signal-to-noise, and minimal crosslinking.
CUT&Tag (Cleavage Under Targets and Tagmentation): An evolution of CUT&RUN. Uses protein A-Tn5 transposase fusion. Upon antibody binding, the loaded Tn5 simultaneously cleaves and inserts sequencing adapters into adjacent DNA. Streamlines library preparation directly on the bead-bound complex.
ChIP-exo: A high-resolution refinement of ChIP-seq. After ChIP, a 5’->3’ exonuclease trims protein-bound DNA to a precise boundary, mapping protein binding sites at near-base-pair resolution.
Re-ChIP (Sequential ChIP): Involves performing two consecutive immunoprecipitations on the same chromatin sample, often with different antibodies. Used to identify genomic regions co-occupied by two different proteins or specific protein isoforms.
Quantitative Comparison: EMSA vs. ChIP Methods
Table 1: Core Comparison of EMSA and Key In Vivo Methods
| Feature | EMSA / Gel Retardation | Standard ChIP (-seq) | CUT&RUN / CUT&Tag |
|---|---|---|---|
| Primary Objective | Confirm direct binding in vitro; assess affinity/specificity. | Map genomic binding sites in vivo; identify target genes. | Map genomic binding sites in vivo with high sensitivity. |
| Context | Cell-free, purified components. | Native chromatin in fixed cells. | Native chromatin in permeabilized cells. |
| Throughput | Low (single loci/probes). | High (genome-wide via seq). | High (genome-wide via seq). |
| Resolution | Binding site defined by probe length (bp). | ~100-200 bp (based on fragment size). | ~10-100 bp (near base-pair for exo variants). |
| Typical Input | Nanograms of recombinant protein. | 10^5 - 10^7 cells. | 10^3 - 10^5 cells. |
| Key Quantitative Output | Binding affinity (Kd), stoichiometry. | Enrichment fold-change, peak locations. | Read counts, peak locations. |
| Time Investment | Fast (1-2 days). | Moderate to Long (3-5 days). | Moderate (2-3 days). |
Table 2: Quantitative Performance Metrics of ChIP Variants (Typical Ranges)
| Method | Recommended Cell Input | Signal-to-Noise Ratio | Mapping Resolution | Hands-on Time | Sequencing Depth Required |
|---|---|---|---|---|---|
| ChIP-seq | 1x10^6 - 1x10^7 | Moderate | 100-300 bp | High | 20-40 million reads |
| ChIP-exo | 1x10^6 - 5x10^6 | High | < 10 bp | Very High | 20-50 million reads |
| CUT&RUN | 5x10^3 - 1x10^5 | High | ~10-100 bp | Low-Moderate | 5-20 million reads |
| CUT&Tag | 5x10^2 - 5x10^4 | High | ~10-100 bp | Low | 5-20 million reads |
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for ChIP and Variants
| Reagent / Material | Function in Experiment | Key Considerations |
|---|---|---|
| Formaldehyde (37%) | Crosslinking agent for standard ChIP. Stabilizes protein-DNA interactions. | Concentration & time critical for efficiency & reverse-crosslinking. |
| Micrococcal Nuclease (MNase) | Enzymatic chromatin shearing (ChIP) or targeted cleavage (CUT&RUN). | Requires titration for optimal fragment size. |
| Protein A/G Magnetic Beads | Solid-phase support for antibody-mediated capture of chromatin complexes. | Superior to agarose beads for washing and automation. |
| High-Affinity, Validated Antibody | Specific recognition of the chromatin protein/epitope of interest. | The single most critical factor; requires ChIP-grade validation. |
| Tn5 Transposase (Loaded) | Simultaneous fragmentation and adapter tagging in CUT&Tag. | Commercial loaded enzymes standardize library prep. |
| Proteinase K | Digests proteins after crosslink reversal to liberate DNA. | Essential for clean DNA recovery. |
| SPRI Beads | Solid-phase reversible immobilization for DNA size selection and cleanup. | Replaces column-based purification for NGS library prep. |
| Sequencing Adapters & Primers | For amplification and sequencing of immunoprecipitated DNA. | Barcoded for multiplexing multiple samples. |
Visualized Workflows and Relationships
Decision Workflow: EMSA vs In Vivo ChIP Methods
Comparative Workflows: Standard ChIP-seq vs CUT&Tag
Evolution of ChIP Method Variants
Within the broader investigation of biomolecular interactions, such as differentiating between EMSA (Electrophoretic Mobility Shift Assay) and gel retardation assays—which both analyze protein-nucleic acid complexes via electrophoresis—the need for quantitative, solution-phase techniques is paramount. SPR and ITC emerge as complementary, label-free methods that provide direct thermodynamic and kinetic data, moving beyond the semi-quantitative, gel-based snapshots provided by EMSA. This guide details their core principles, protocols, and synergistic application in modern research and drug development.
Core Principles and Complementary Data
Surface Plasmon Resonance (SPR) measures real-time biomolecular interactions by detecting changes in the refractive index near a sensor surface. One molecule (ligand) is immobilized, and the binding partner (analyte) flows over it. The resulting sensorgram provides kinetic data (association rate k~a~, dissociation rate k~d~) and an equilibrium dissociation constant (K~D~).
Isothermal Titration Calorimetry (ITC) directly measures heat released or absorbed during binding. A titration of one molecule into another provides thermodynamic data: binding constant (K~a~), enthalpy change (ΔH), entropy change (ΔS), and stoichiometry (n).
Together, they furnish a complete picture: SPR reveals how fast a complex forms and dissociates, while ITC explains why binding occurs by detailing the driving forces.
Table 1: Comparative Output of SPR and ITC
| Parameter | SPR | ITC | Complementary Insight |
|---|---|---|---|
| Primary Output | Kinetic rates, K~D~ (nM-μM) | ΔH, ΔS, K~a~, n | Kinetics + Thermodynamics |
| Sample Consumption | Low (μg ligand) | Moderate-High (mg) | SPR for screening, ITC for lead validation |
| Throughput | Medium-High | Low | SPR for multi-condition analysis |
| Label Required? | No (but surface immobilization) | No | Both are label-free |
| Key Assumption | Mass transport not limiting; minimal surface artifacts | Heat change is solely from binding | Validate K~D~/K~a~ agreement between techniques |
Detailed Experimental Protocols
Protocol 1: SPR for Protein-DNA Interaction Analysis
(As relevant to EMSA-related studies)
1. Sensor Chip Preparation:
- Use a carboxymethylated dextran (CM5) sensor chip.
- Activate the surface with a 1:1 mixture of 0.4 M EDC and 0.1 M NHS for 7 minutes.
- Dilute the DNA oligonucleotide (ligand) in sodium acetate buffer (pH 5.0) to 50 μg/mL. Inject until ~100-200 Response Units (RU) are immobilized.
- Deactivate with 1 M ethanolamine-HCl (pH 8.5) for 7 minutes.
- Create a reference flow cell with activation/deactivation only.
2. Binding Kinetics Experiment:
- Use HBS-EP+ buffer (10 mM HEPES, 150 mM NaCl, 3 mM EDTA, 0.05% v/v Surfactant P20, pH 7.4) as running buffer.
- Dilute the protein (analyte) in running buffer in a 2-fold series across 6 concentrations (e.g., from K~D~/3 to 3K~D~).
- Inject each concentration for 120-180s (association phase) at 30 μL/min.
- Monitor dissociation for 300-600s.
- Regenerate surface with a 30s pulse of 1M NaCl if needed.
3. Data Analysis:
- Subtract reference flow cell data.
- Fit double-referenced data to a 1:1 Langmuir binding model to extract k~a~, k~d~, and K~D~ (K~D~ = k~d~/k~a~).
Protocol 2: ITC for Binding Thermodynamics
1. Sample Preparation:
- Exhaustively dialyze both the protein (in cell) and DNA (in syringe) into identical buffers (e.g., 20 mM phosphate, 150 mM NaCl, pH 7.0). Buffer matching is critical.
- Degas both samples for 10 minutes prior to loading.
2. Titration Experiment:
- Load the protein solution (typically 10-50 μM) into the sample cell (1.4 mL).
- Load the DNA solution (typically 5-10x more concentrated) into the syringe.
- Set temperature to 25°C.
- Program: Initial 0.4 μL injection (discarded), followed by 18-25 injections of 1.5-2.0 μL each, spaced 180s apart with constant stirring at 750 rpm.
3. Data Analysis:
- Integrate raw heat peaks per injection.
- Subtract heats of dilution (from control titration of DNA into buffer).
- Fit the corrected isotherm to a single-site binding model to obtain K~a~, ΔH, and n. Calculate ΔG (ΔG = -RT lnK~a~) and ΔS (ΔS = (ΔH - ΔG)/T).
Visualizing Workflow and Data Integration
Title: SPR and ITC Complementary Analysis Workflow
Title: From EMSA Screening to SPR/ITC Quantification
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for SPR & ITC Experiments
| Item | Function | Critical Consideration |
|---|---|---|
| Biacore Series S Sensor Chip (CM5) | Gold surface with carboxymethylated dextran matrix for ligand immobilization. | Industry standard for SPR; choose chip type (e.g., NTA for His-tag) based on application. |
| Amine Coupling Kit (EDC, NHS, Ethanolamine) | Chemicals for covalent immobilization of ligands via primary amines. | Must be fresh; aliquots should be stored desiccated at -20°C. |
| High-Purity HBS-EP+ Buffer | Standard running buffer for SPR; minimizes non-specific binding. | Use filtered, degassed buffer to prevent air bubbles and system clogging. |
| MicroCal ITC Disposable Syringe | Precision syringe for titrant delivery in ITC. | Must be meticulously cleaned and rinsed with filtered buffer to prevent contamination. |
| Dialysis Cassettes (3.5 kDa MWCO) | For exhaustive buffer exchange of ITC samples. | Essential for perfect buffer matching to avoid heat artifacts from dilution. |
| OriginLab or MicroCal PEAQ-ITC Analysis Software | For integrating ITC peaks and nonlinear curve fitting. | Accurate baseline assignment is crucial for correct thermodynamic parameters. |
| ProteOn or Biacore Evaluation Software | For processing SPR sensorgrams and kinetic fitting. | Requires careful referencing and model selection for reliable kinetics. |
Within the broader thesis of distinguishing EMSA from gel retardation assays, it is critical to recognize that "Electrophoretic Mobility Shift Assay" (EMSA) is the modern, specific term that falls under the historical umbrella of "gel retardation assays." The contemporary distinction lies not between these two terms but between the qualitative/semi-quantitative nature of traditional EMSA and truly quantitative binding methodologies. This guide provides a technical framework for selecting the appropriate assay based on research objectives.
Core Principles and Quantitative Output
EMSA (Electrophoretic Mobility Shift Assay): EMSA separates protein-nucleic acid complexes from free nucleic acid via native gel electrophoresis. The shift in mobility indicates binding. While intensity of bands can be quantified via densitometry, the assay is inherently semi-quantitative due to factors like complex dissociation during electrophoresis and staining inefficiencies.
Quantitative Binding Assays: These include techniques like Surface Plasmon Resonance (SPR), Isothermal Titration Calorimetry (ITC), and Fluorescence Polarization/Anisotropy (FP/FA). They measure binding in real-time in solution, providing direct measurements of association ((ka)), dissociation ((kd)) rates, and equilibrium constants ((K_D)).
Quantitative Data Comparison
Table 1: Comparative Analysis of Binding Assays
| Parameter | EMSA / Gel Retardation | SPR (Biacore) | ITC | Fluorescence Anisotropy |
|---|---|---|---|---|
| Primary Output | Shifted band (visual), % complex formation | Resonance Units (RU) vs. time, sensograms | Heat flow (μcal/sec) vs. time | Polarization/Anisotropy (mP) |
| Quantitative Readout | Semi-quantitative (densitometry) | Directly quantitative | Directly quantitative | Directly quantitative |
| Key Metrics | Apparent (K_D) (with caveats) | (ka), (kd), (K_D) (kinetic & equilibrium) | (ΔH), (ΔS), (n) (stoichiometry), (K_D) (thermodynamic) | (K_D) (equilibrium) |
| Typical (K_D) Range | nM - μM | pM - mM | nM - μM | nM - μM |
| Throughput | Low to medium | Medium | Low | High (plate-based) |
| Sample Consumption | Moderate to high | Low (ligand immobilized) | High (for precise ΔH) | Low |
| Real-time Monitoring | No (endpoint assay) | Yes | Yes | Yes (endpoint or kinetic) |
| Native Condition Support | High (native gel) | Medium (one partner immobilized) | High (in solution) | High (in solution) |
Experimental Protocols
Protocol 1: Standard EMSA for Transcription Factor Binding
Objective: To detect and semi-quantify the binding of a nuclear protein extract to a radiolabeled DNA probe containing a consensus sequence.
Materials:
- Binding Buffer: 10 mM Tris, 50 mM KCl, 1 mM DTT, 2.5% glycerol, 5 mM MgCl₂, 0.05% NP-40, pH 7.5.
- Poly(dI:dC): Non-specific competitor DNA.
- ³²P-end-labeled DNA probe: 20,000-50,000 cpm per reaction.
- Nuclear extract or purified protein.
- Native Polyacrylamide Gel: 4-6% acrylamide (29:1 acrylamide:bis), 0.5X TBE.
- Electrophoresis: Run in 0.5X TBE buffer at 100V, 4°C.
- Detection: Dry gel and expose to phosphorimager screen.
Procedure:
- Prepare 20 μL binding reactions: 1X binding buffer, 1 μg poly(dI:dC), labeled probe (1-10 fmol), and protein extract (2-10 μg).
- Incubate at room temperature for 20-30 minutes.
- Load onto pre-run native gel immediately (do not add loading dye with SDS or EDTA).
- Run at 100V, 4°C until the free probe has migrated ~2/3 down the gel.
- Transfer gel to blotting paper, dry under vacuum, and visualize via autoradiography or phosphorimaging.
Protocol 2: Determining (K_D) by Fluorescence Anisotropy (Quantitative)
Objective: To determine the equilibrium dissociation constant ((K_D)) for a protein-fluorescently-labeled DNA interaction in solution.
Materials:
- Assay Buffer: Suitable for protein activity (e.g., Tris, NaCl, DTT, BSA).
- Tracer: DNA oligonucleotide labeled at 5' or 3' with a fluorophore (e.g., FAM, TAMRA).
- Purified Protein: Serial dilutions in assay buffer.
- Black 384-well plate.
- Plate reader with fluorescence polarization/anisotropy capability.
Procedure:
- Prepare a 2X serial dilution of the protein in assay buffer across 12 points, covering a concentration range from below to above the expected (K_D).
- In each well of the plate, mix equal volumes of the protein dilution and a fixed, low concentration (typically ≤ (K_D)) of the fluorescent tracer.
- Incubate in the dark for 30-60 minutes to reach equilibrium.
- Measure anisotropy (r) for each well.
- Plot anisotropy vs. log[protein]. Fit data to a 1:1 binding isotherm model: ( r = r{free} + (r{bound} - r{free}) * ( [P] / (KD + [P]) ) ) where [P] is protein concentration.
Decision Pathway: EMSA vs. Quantitative Assays
Decision Tree for Binding Assay Selection
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagent Solutions for Nucleic Acid-Protein Binding Studies
| Reagent / Material | Function / Purpose | Typical Example / Specification |
|---|---|---|
| Purified Recombinant Protein | The binding partner of interest; requires high purity and activity for quantitative assays. | >95% purity, confirmed activity, in stable storage buffer. |
| Labeled Nucleic Acid Probe | The detectable binding target. For EMSA, often ³²P-γ-ATP end-labeled. For FA, fluorophore-labeled (FAM, Cy3, TAMRA). | 20-50 bp DNA/RNA, HPLC-purified, specific activity defined. |
| Non-specific Competitor DNA | Reduces non-specific binding of proteins to the probe, critical for EMSA with crude extracts. | Poly(dI:dC), sheared salmon sperm DNA. |
| Native Gel Matrix | For EMSA: separates complex from free probe based on size/sharge without denaturation. | 4-8% acrylamide/bis (29:1 or 37.5:1), 0.5X TBE buffer. |
| SPR Chip / Sensor Surface | For SPR: immobilizes one binding partner (ligand) to monitor interaction with analyte in flow. | CM5 dextran chip (for amine coupling), NTA chip (for His-tagged proteins). |
| ITC Cell & Syringe | For ITC: contains the sample and injectant. Requires meticulous cleaning and degassing of solutions. | 200 μL sample cell, 40 μL injection syringe. |
| Fluorescent Tracer | For FA/FP: the basis of the homogenous, solution-phase signal. Must be bright and photostable. | 5'-FAM-labeled oligonucleotide, kept protected from light. |
| Anisotropy Plate Reader | For FA/FP: measures the polarization of emitted light, which increases upon protein binding to the tracer. | Instrument capable of measuring fluorescence polarization (mP units). |
Workflow Visualization: From Experiment to Data
EMSA vs Quantitative Assay Workflow Comparison
The choice between EMSA and a quantitative binding assay is fundamental to research design. EMSA remains unparalleled for its simplicity, ability to handle complex samples, and visual confirmation of complex formation—making it ideal for initial discovery and validation. However, for lead optimization in drug discovery, mechanistic studies requiring kinetic and thermodynamic parameters, or any scenario demanding rigorous affinity comparisons, quantitative solution-phase assays like SPR, ITC, and FA are indispensable. Within the thesis of EMSA vs. gel retardation research, understanding this qualitative-to-quantitative continuum equips researchers to apply the right tool for their specific stage of inquiry.
1. Introduction: Framing EMSA within Broader Electrophoretic Mobility Research
Electrophoretic Mobility Shift Assay (EMSA) and gel retardation assay are often used synonymously, yet a technical thesis distinguishes them. While both rely on the principle that protein-nucleic acid complexes migrate slower than free nucleic acids in gels, "gel retardation" is a broader term encompassing various complexation events. EMSA is a specific, refined application focused on detecting sequence-specific binding of proteins to DNA or RNA, typically employing labeled probes and controlled competition experiments. This whitepaper focuses on EMSA as a quantitative in vitro tool and details strategies to transcend its inherent limitation: proving that observed binding correlates with cellular function and phenotype.
2. From In Vitro Binding to Cellular Relevance: An Integrated Workflow
The core challenge is connecting a biophysical interaction measured in vitro to a biological outcome. The following workflow outlines a multi-validation approach.
Diagram 1: Integrated workflow from EMSA to phenotype.
3. Quantitative EMSA: Protocols and Data Analysis
3.1 Detailed Protocol for Quantitative EMSA
- Probe Labeling: Use T4 Polynucleotide Kinase and [γ-³²P]ATP or a chemiluminescent/fluorescent tag. Purify labeled probe via spin column.
- Binding Reaction (20 µL):
- Nuclear extract (5-10 µg) or purified protein (1-100 nM).
- Labeled probe (10-20 fmol).
- Poly(dI-dC) (1-2 µg) as non-specific competitor.
- Binding buffer (10 mM HEPES pH 7.9, 50 mM KCl, 1 mM DTT, 2.5 mM MgCl₂, 10% glycerol, 0.05% NP-40).
- Incubate 20-30 min at room temperature.
- Competition EMSA: Include unlabeled wild-type or mutant oligonucleotide (10-1000x molar excess) in the reaction.
- Supershift: Add 1-2 µg of specific antibody.
- Electrophoresis: Load on pre-run 4-6% native polyacrylamide gel (0.5X TBE, 4°C). Run at 100V until free probe migrates ~2/3 down.
- Detection: Autoradiography, phosphorimaging, or chemiluminescence.
3.2 Data Quantification and Table Quantify band intensity (free vs. bound) using ImageJ or dedicated software. Fit data to determine equilibrium dissociation constant (Kd) for purified proteins.
Table 1: Quantitative EMSA Data for Hypothetical Transcription Factor (TF-X)
| Experiment Type | Condition | % Probe Bound (Mean ± SD) | Derived Metric | Interpretation |
|---|---|---|---|---|
| Titration (Purified TF-X) | 1 nM TF-X | 15 ± 3% | Kd = 5.2 nM | High in vitro affinity for consensus sequence. |
| 10 nM TF-X | 65 ± 5% | |||
| Specificity Competition | 100x unlabeled WT | 8 ± 2% (vs. 65%) | IC50 (WT) ≈ 25x fold | Binding is sequence-specific. |
| 100x unlabeled Mutant | 60 ± 4% (vs. 65%) | IC50 (Mut) > 500x fold | ||
| Cellular Extract EMSA | Control Nuclear Extract | 22 ± 4% | Baseline activity in cells. | TF-X is active and binding in vitro. |
| siRNA-TF-X Extract | 5 ± 2% | >75% reduction | Confirms complex identity. | |
| Antibody Supershift | Control IgG | No shift | Complex identity confirmed. | TF-X is part of the DNA-protein complex. |
| α-TF-X IgG | Complete supershift |
4. Correlative & Causal Cellular Experiments
4.1 Protocol: CRISPR-Cas9 Mediated Binding Site Mutation
- Design: Use CRISPR design tools to create gRNAs flanking the EMSA-validated binding site in the genomic locus of interest.
- Transfection: Deliver Cas9/gRNA RNP or plasmid into cells via nucleofection.
- Screening: Isolate clones and sequence the target region. Select homozygous mutants.
- Validation: Perform EMSA with nuclear extract from mutant cells and the probe matching the genomic sequence to confirm loss of complex formation in vitro.
4.2 Protocol: Chromatin Immunoprecipitation (ChIP-qPCR)
- Crosslink: Treat cells with 1% formaldehyde for 10 min.
- Lysis & Sonication: Lyse cells and shear chromatin to 200-500 bp fragments.
- Immunoprecipitation: Incubate with anti-TF-X antibody or isotype control, then pull down with protein A/G beads.
- Reverse Crosslinks & Analyze: Elute, reverse crosslinks, purify DNA, and analyze by qPCR using primers for the putative binding site and a control region.
Table 2: Correlating EMSA Data with Cellular Functional Assays
| Cellular Experiment | Experimental Group | Key Readout | Correlation with EMSA |
|---|---|---|---|
| ChIP-qPCR | Wild-type cells | Fold enrichment vs. control IgG | Strong enrichment should align with high % probe bound in EMSA. |
| CRISPR binding-site mutant | Loss of enrichment | Should mirror loss of complex in EMSA with mutant probe. | |
| RT-qPCR of Target Gene | TF-X Overexpression | Increased target mRNA | Confirms TF-X binding from EMSA is functionally activating. |
| TF-X Knockdown | Decreased target mRNA | Strengthens causal link between binding and regulation. | |
| Phenotypic Assay | TF-X Knockdown | e.g., Reduced proliferation | A phenotype suggests the EMSA-detected pathway is functionally significant. |
| Pharmacologic inhibitor | Phenotype mimicking KD | If inhibitor disrupts EMSA complex, links binding to phenotype. |
5. The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for Integrated EMSA Studies
| Reagent / Material | Function & Importance |
|---|---|
| Chemiluminescent Nucleic Acid Labeling Kit | Non-radioactive, sensitive probe labeling for EMSA. Safer and stable. |
| High-Quality Nuclear Extraction Kit | Prepares active transcription factors from cellular samples for EMSA validation. |
| Recombinant Purified Protein | Essential for quantitative Kd determination by EMSA titration. |
| Custom Wild-type & Mutant Oligonucleotides | For probe generation and unlabeled competitors. Critical for proving specificity. |
| Specific Antibody (for Supershift) | Confirms protein identity in the EMSA complex. Must be capable of recognizing native protein. |
| ChIP-Grade Antibody | Validated for chromatin immunoprecipitation. Different epitope requirement than supershift. |
| CRISPR-Cas9 Gene Editing System | To create causal mutations in genomic binding sites, linking in vitro data to cellular function. |
| Dual-Luciferase Reporter Assay System | Validates if EMSA-detected binding site confers regulatory activity in living cells. |
6. Pathway Integration and Data Synthesis
To fully integrate data, map EMSA-validated interactions onto known signaling or transcriptional pathways.
Diagram 2: Pathway context for EMSA-validated binding.
7. Conclusion
Effective integration requires moving from the qualitative "band shift" to quantitative binding parameters, and systematically testing the functional necessity and sufficiency of that interaction through cellular genetics and phenotyping. Within the broader thesis of electrophoretic mobility assays, EMSA's unique power lies in its biochemical specificity, which, when used as a foundation for causal cellular experiments, robustly bridges in vitro biophysics to in vivo biology and phenotype.
The electrophoretic mobility shift assay (EMSA), often colloquially termed the "gel retardation assay," is a cornerstone technique for studying protein-nucleic acid interactions. Within the context of a broader thesis on the Difference between EMSA and gel retardation assay research, it is critical to establish that while the terms are frequently used interchangeably, EMSA represents a refined and standardized application of the core gel retardation principle. This whiteprame focuses on the sophisticated use of EMSA in modern drug discovery, specifically for identifying compounds that disrupt pathogenic interactions, a process that leverages the quantitative and qualitative improvements of EMSA over classical retardation assays.
EMSA Fundamentals and Quantitative Data
EMSA detects complex formation by observing a reduction in the electrophoretic mobility of a fluorescently or radioactively labeled nucleic acid probe upon binding to a protein. In drug screening, a decrease in the shifted band intensity in the presence of a compound indicates potential inhibition.
Table 1: Key Quantitative Parameters in EMSA-Based Screening
| Parameter | Typical Range / Value | Significance in Screening |
|---|---|---|
| Protein Concentration | 1-100 nM | Must be titrated to achieve ~70-80% probe binding for robust inhibition signals. |
| Nucleic Acid Probe Conc. | 0.1-1 nM (labeled) | Kept significantly below protein concentration to ensure binding equilibrium. |
| Incubation Time (Binding) | 20-30 min @ 4-25°C | Allows complex formation to reach equilibrium. |
| Electrophoresis Conditions | 4-10°C, 80-120 V, 5% TBE Polyacrylamide gel | Non-denaturing conditions preserve complexes. |
| IC₅₀ Determination (from EMSA) | nM to µM range | Concentration of inhibitor that reduces complex formation by 50%. |
| Z'-Factor for HTS EMSA | >0.5 | Statistical parameter indicating robustness of assay for high-throughput screening. |
Detailed Experimental Protocol for Inhibitor Screening
Protocol: EMSA for Primary Screening of Inhibitors
Objective: To identify small molecules that disrupt the binding of a pathogenic protein (e.g., viral protease-integrase, bacterial transcription factor) to its target DNA/RNA sequence.
I. Materials & Reagent Preparation
- Binding Buffer (10X Stock): 100 mM Tris-HCl (pH 7.5), 500 mM KCl, 50 mM MgCl₂, 10 mM DTT, 50% Glycerol. Store at -20°C.
- Protein: Recombinant, purified pathogenic protein. Aliquot and store at -80°C.
- Probe: Double-stranded DNA or RNA oligonucleotide containing the target sequence. Label with IRDye 800CW (fluorescence) or [γ-³²P]ATP (radioactivity). Purify via gel filtration.
- Competitor DNA: Unlabeled nonspecific DNA (e.g., poly(dI-dC)) to suppress nonspecific binding.
- Test Compounds: Small molecule library compounds dissolved in DMSO. Final DMSO concentration in assay ≤1%.
- Gel: 6% non-denaturing polyacrylamide gel (29:1 acrylamide:bis) in 0.5X TBE. Pre-run for 30-60 min.
- Equipment: Gel electrophoresis apparatus, fluorescence imager or phosphorimager.
II. Procedure
- Complex Formation: For each 20 µL reaction, combine:
- 2 µL 10X Binding Buffer
- 1 µL poly(dI-dC) (1 µg/µL)
- X µL Protein (final conc. determined via titration)
- Y µL Test Compound (or DMSO control)
- Nuclease-free water to 19 µL. Incubate at room temperature for 15 min.
- Add Probe: Add 1 µL of labeled probe (0.1 nM final). Mix gently.
- Final Incubation: Incubate at reaction temperature (e.g., 30°C) for 25 min.
- Electrophoresis: Load entire reaction onto pre-run gel. Run in 0.5X TBE at 100 V, 4°C for 60-90 min (until dye front migrates appropriately).
- Detection: Image gel using appropriate channel (fluorescence) or expose to phosphor screen (radioactivity).
III. Analysis Quantify band intensities for free probe and protein-probe complex. Calculate % bound probe for each reaction. Compounds showing significant reduction in % bound versus DMSO control are candidate inhibitors.
Visualization of Workflows and Pathways
Title: EMSA-based inhibitor screening workflow
Title: EMSA evolution within gel retardation research
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for EMSA-Based Inhibitor Screening
| Item | Function & Rationale |
|---|---|
| Recombinant Pathogenic Protein | High-purity, active protein is essential. Often tagged (e.g., His-tag) for purification. Activity must be validated prior to screening. |
| Fluorescently-Labeled Nucleic Acid Probes (e.g., IRDye 800CW, Cy5) | Safer and more convenient than radioactive labels, enabling rapid imaging. Must be HPLC-purified. |
| Non-specific Competitor DNA (e.g., poly(dI-dC), salmon sperm DNA) | Critical for blocking non-sequence-specific protein-nucleic acid interactions, improving signal-to-noise. |
| Optimized Non-denaturing PAGE System | Pre-cast gels or standardized recipes ensure reproducible migration of complexes. Cold electrophoresis buffers prevent complex dissociation. |
| High-Sensitivity Gel Imager | Fluorescence scanners or phosphorimagers capable of detecting low nanomolar probe concentrations are required for quantification. |
| Microplate-Compatible EMSA Kits | Commercial kits provide standardized buffers and protocols adapted for medium- to high-throughput screening in 96- or 384-well formats. |
| Negative Control Probe (Mutated sequence) | Validates that protein binding and compound inhibition are sequence-specific. |
| Reference Inhibitor (Known weak binder) | Serves as a positive control for inhibition in each screening run, ensuring assay performance. |
Conclusion
EMSA, or the gel retardation assay, remains a foundational, versatile, and accessible technique for studying nucleic acid-protein interactions. While its core principle is simple, mastery requires careful attention to methodological detail, systematic troubleshooting, and an understanding of its place within a larger validation framework. As we have explored, its value lies not only in detecting binding events but also in providing a platform for competitive and supershift analyses. For modern researchers and drug developers, EMSA is often the critical first step in characterizing interactions, but its findings should be strengthened by complementary in vivo and quantitative in vitro techniques. Future directions point towards increased automation, higher sensitivity with novel detection chemistries, and integration with computational models of binding dynamics, ensuring EMSA's continued relevance in mechanistic biology and therapeutic discovery.